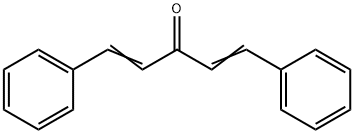
Synthesis and Conformations of Dibenzylideneacetone
Dibenzylideneacetone appears as pale yellow needle-like crystals under normal temperature and pressure. It exhibits excellent fluorescence properties and good chemical stability. It is insoluble in water but soluble in ethanol, acetone, and chloroform. Dibenzylideneacetone can be used as a ligand in the preparation of palladium catalysts, which are widely applied in catalytic hydrogenation, coupling reactions, carbonylation, alkyne cyclotrimerization, and other processes. Examples include palladium-catalyzed reactions such as the Negishi reaction (organozinc compounds with halides), the Stille reaction (organotin compounds with halides), the Suzuki reaction (organoboron compounds with halides), and the Heck reaction (halides with olefins).
Synthesis
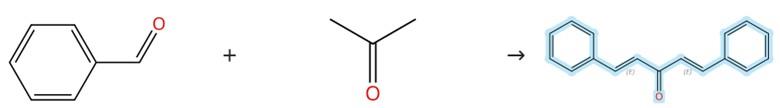
Figure1: Synthesis of Dibenzylideneacetone
To a stirred ethanolic solution (5 mL) containing a benzaldehyde derivative (2.702 mmol), acetone (1.35 mmol) is introduced, followed by the addition of an aqueous solution of LiOH·H₂O (10 mol%, 0.27 mmol). The resulting mixture is allowed to stir at room temperature for a period of 30 minutes, during which time the progress of the reaction is meticulously monitored by thin-layer chromatography. Upon confirmation of completion, the reaction is carefully quenched by the addition of an excess of water (30 mL), leading to the formation of a precipitate, which is subsequently isolated by filtration. The obtained crude product is then subjected to purification via recrystallization using a mixture of ethyl acetate and hexane to give Dibenzylideneacetone. [1]
Conformations
The conformational analysis of dibenzylideneacetone (DBA) was conducted using IR spectroscopy, revealing a triplet C=O band that indicates the coexistence of three conformers—namely s-cis, cis; s-cis, trans; and nonplanar s-trans, trans—contrary to earlier studies that reported only two conformers. The relative proportions of these conformers vary with solvent polarity: in less polar solvents, the order is s-cis, trans > nonplanar s-trans, trans > s-cis, cis, while in more polar solvents, the sequence shifts to nonplanar s-trans, trans > s-cis, trans > s-cis, cis. This conformational analysis of DBA is particularly significant due to its ability to form a novel series of zerovalent transition metal complexes, a property stemming from its existence in multiple isomeric forms. The presence of two single bonds between the olefin and carbonyl group provides two degrees of rotational freedom, enabling three possible conformations for dibenzylideneacetone: s-cis, cis; s-cis, trans; and s-trans, trans. [2]
UV absorption spectra
The UV absorption spectra of dibenzylideneacetone (DBA) were measured in various media—including cyclohexane, ethanol, ethanol-methanol, and a polyethylene matrix—at room temperature and at 101 K. The experimental results, along with those calculated using the PPP method, indicate that dibenzylideneacetone adopts a twisted cis,cis-conformation at room temperature when embedded in the polyethylene matrix, whereas it assumes a near-planar conformation at low temperature. At room temperature, the torsion angles around the two single bonds between the olefin and carbonyl groups in dibenzylideneacetone are estimated to be approximately 22°. The effects of light irradiation on the molecular structure of dibenzylideneacetone were also investigated, and the polarization of each band in its polarized absorption spectrum was determined. For instance, the first π*←π band of the planar cis,cis-conformer of dibenzylideneacetone, observed at 351 nm, is polarized along the long molecular axis. Additionally, MIND0/2 calculations were performed to elucidate the equilibrium conformations of the free dibenzylideneacetone molecule. [3]
Hydrogenation reaction
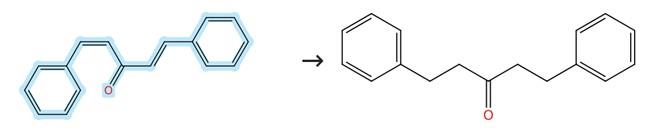
Figure2: Hydrogenation reaction of Dibenzylideneacetone
Dibenzylideneacetone (1.6 mmol) along with Raney Ni (10 wt.%) are added to a 100 mL high-pressure reactor equipped with a magnetic stirrer, which is subsequently charged with DCM (8 mL) and subjected to hydroge by introducing H₂ (1.0 MPa) at the inlet and venting three times to ensure an inert atmosphere. The reactor is then pressurized with hydrogen to 1.0 MPa, and the mixture is vigorously stirred at 30°C for 3 hours, with the temperature maintained using a heating mantle. After the reaction is complete, the hydrogen is carefully released in a fume hood, and the mixture is filtered to remove the Raney Ni catalyst. The filtrate is washed with ethyl acetate (3 × 10 mL), and the combined organic layers are dried over anhydrous sodium sulfate, filtered, and concentrated to dryness under reduced pressure. [4]
Reference
[1] Andati, Reu E.; Ovicidal Activity of 2-Hydroxy-4-Methoxybenzaldehyde, Derivatives and Structural Analogs on Anopheles gambiae Eggs, Journal of Chemistry 2025, 2025, 3387088
[2] Venkateshwarlu G, Subrahmanyam B. Conformations of dibenzylideneacetone: An IR spectroscopic study[C]//Proceedings of the Indian Academy of Sciences-Chemical Sciences. New Delhi: Springer India, 1987, 99: 419-424.
[3] Hoshi T, Kawashima T, Okubo J, et al. Conformations and electronic structures of dibenzylideneacetone[J]. Journal of the Chemical Society, Perkin Transactions 2, 1986 (8): 1147-1150.
[4] Zhang, Yu-kun; et al, Solvent-Controlled and Highly Chemoselective Reduction of α,β-Unsaturated Ketones and Aldehydes, Journal of Organic Chemistry (2025), 90(16), 5704-5709.
You may like
See also
Lastest Price from Dibenzylideneacetone manufacturers

US $0.00/kg2025-03-10
- CAS:
- 538-58-9
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 500 kgs