Synthesis and Epoxidation Reaction of 2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone
2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone is an important intermediate in pharmaceutical chemistry, typically appearing as a white to pale yellow solid powder under ambient conditions. In the field of pharmaceutical production, 2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone is primarily used in the synthesis of the voriconazole racemate. Voriconazole is a broad-spectrum triazole antifungal agent for the treatment of invasive fungal infections (IFI); it works mainly by inhibiting cytochrome P450-dependent enzymes, thereby interfering with ergosterol synthesis in fungal cells. The resulting ergosterol deficiency inhibits fungal cell growth, thus exerting its antifungal effect.
Synthesis
Research has publicly reported a method for preparing 2,4-difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone. Using 2-chloro-2',4'-difluoroacetophenone as the starting material, the reaction with 3-chloro-1,2,4-triazole yields 2-(3-chloro-1H-1,2,4-triazol-1-yl)-1-(2,4-difluorophenyl)ethanone, which is then subjected to palladium-on-carbon hydrogenation dehalogenation to obtain the final product 2,4-difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone. The preparation of 2,4-difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone via this method features simple steps, convenient operation, and low economic cost, making it suitable for industrial production. This approach is expected to deliver favorable social and economic benefits, with considerable potential for economic value. [1]
Preparation of alkylated piperazine-azole hybrids
Azole antifungal drugs are widely used to treat fungal infections, but they have led to the emergence of drug resistance. Therefore, developing azole derivatives that can both combat existing drug-resistant fungal strains and circumvent resistance is of great significance. Research has reported that alkylated piperazine-azole hybrids exhibit significantly better antifungal activity than fluconazole (FLC), particularly showing stronger inhibitory effects against non-albicans Candida and Aspergillus strains. Therefore, researchers designed and synthesized a series of azole analogs by conjugating the FLC skeleton with triphenylphosphonium cation (TPP⁺) through alkyl linkers. In this design, the piperazine structural unit was used to replace the original 1,2,4-triazole group on the α-carbon linked to the difluorophenyl ring in FLC. The synthesis route started with the commercially available 2,4-difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone, the carbonyl group of which underwent epoxidation with trimethylsulfoxonium iodide under strong basic conditions to form the epoxide intermediate. For piperazine-substituted azole analogs, the epoxide was reacted with piperazine to generate an intermediate, which was then treated with various alkyl dibromides under basic conditions to afford brominated intermediates. Finally, these brominated intermediates were coupled with another intermediate prepared according to literature methods to yield TPP-conjugated products. [2]
Epoxidation Reaction
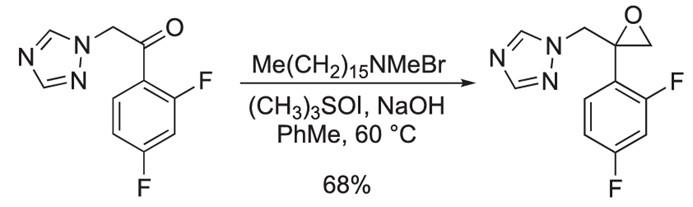
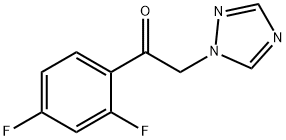
Figure1: Epoxidation Reaction of 2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone
To a stirred solution of 2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone (5.00 g, 22.4 mmol) in toluene (30.0 mL), trimethylsulfoxonium iodide (5.40 g, 26.9 mmol), hexadecyltrimethylammonium bromide (815 mg, 2.25 mmol), and 7.50 mL of 20.0% (v/v) sodium hydroxide were added at room temperature. The reaction mixture was stirred at 60 C for 4 h, and the reaction was monitored by TLC. When finished, the reaction was quenched with water, and extrated with dichloromethane. The combined extracts were washed with brine, dried over MgSO4 , f iltered and concentrated under reduced pressure to give the crude product, which was purified by flash chromatography to provide com pound 3 (3.60 g, 68.0%) as a yellow gummy oil. [2]
Benzylation Reaction
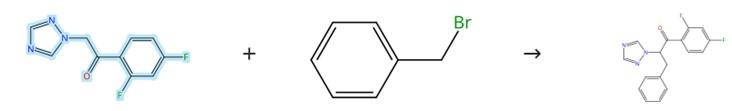
Figure2: Benzylation Reaction of 2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone
Add a solution of the 2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone (6 mmol) in acetonitrile (10 ml) to a suspension of NaH (8 mmol) in acetonitrile (30 ml), and stir the resulting mixture at room temperature for 1 hour. Then, add a solution of the benzyl bromide reactant (6 mmol) in acetonitrile (10 ml) dropwise, and reflux the mixture for 24 hours. After cooling the reaction mixture to room temperature, evaporate the solvent under reduced pressure. Wash the crude product with water (50 ml), separate the aqueous layer, and extract the mixture with dichloromethane (3 × 30 ml). Dry the combined organic layers over Na₂SO₄, and finally purify the crude product by column chromatography on silica gel using ethyl acetate and petroleum ether as the eluent. [3]
Reference
[1] Lu G Y, Zhou Y H. A preparation method of 2',4'-difluoro-2-[1-(1H-1,2,4-triazolyl)]acetophenone: CN202010335499.7[P].
[2] Wang X, et al. Azole-triphenylphosphonium conjugates combat antifungal resistance and alleviate the development of drug-resistance[J]. Bioorganic Chemistry, 2021, 110: 104771.
[3] 1,2,4-Triazole derivatives as novel and potent antifungal agents: Design, synthesis and biological evaluation, Sadeghian, Sara; et al, Journal of Molecular Structure (2023), 1271, 134039
You may like
See also
Lastest Price from 2,4-Difluoro-alpha-(1H-1,2,4-triazolyl)acetophenone manufacturers

US $0.00/KG2025-04-15
- CAS:
- 86404-63-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg

US $60.00/kg2025-04-15
- CAS:
- 86404-63-9
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 5000kg/week