Synthesis and Esterification Reaction of 2-Hydroxyphenylacetic acid
2-Hydroxyphenylacetic acid is a white solid powder under ambient temperature and pressure, characterized by notable acidity and excellent chemical stability. While insoluble in water, it exhibits certain solubility in alkaline aqueous solutions. Primarily utilized as a basic raw material in agricultural insecticides and fine chemical production, 2-Hydroxyphenylacetic acid has been reported in research as a key intermediate for synthesizing methoxyacrylate‑type fungicides, such as azoxystrobin, and their active derivatives.

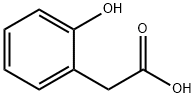
Figure1: Picture of 2-Hydroxyphenylacetic acid
Synthesis
Method 1
A preparation method for 2-Hydroxyphenylacetic acid has been publicly reported. In this method, 2‑chlorophenylacetic acid, 40 wt% aqueous sodium hydroxide solution, and copper(I) chloride are charged into a pressure reactor in a molar ratio of 0.95‑1.05 : 3.71‑3.81 : 0.03‑0.13. After the addition, the mixture is heated to 160‑180°C, then allowed to rise naturally to 230‑240°C and maintained at this temperature for 8 hours. Upon completion of the reaction, the reaction mixture is cooled below 100°C, diluted with water, and stirred uniformly. The mixture is then filtered under pressure and transferred to an acidification vessel. While controlling the temperature at 45‑55°C, the solution is acidified with 30 wt% dilute sulfuric acid to a pH of 2‑3. After cooling to 35‑40°C, additional water is added, and the mixture is stirred for 30‑40 minutes before being centrifuged. The crude solid obtained from centrifugation is transferred to a washing vessel, washed with water at a mass ratio of crude product to water of 1:5, stirred for 30 minutes, and centrifuged to yield the final product, namely 2‑hydroxyphenylacetic acid. The preparation method described in the present invention offers simple operation, time‑saving advantages, significantly improves product yield, and is environmentally friendly, making it suitable for the large‑scale production of 2-Hydroxyphenylacetic acid. [1]
Method 2
Marine fungi belonging to the genera Aspergillus, Penicillium, Cladosporium, andBionectria catalyzed the biotransformation of phenylacetonitrile to 2 hydroxyphenylacetic acid. Eight marine fungi, selected and cultured with phenylacetonitrile in liquid mineral medium, catalyzed it quantitative biotransformation to 2-hydroxyphenylacetic acid. In this study, the nitrile group was firstly hydrolysed, and then, the aromatic ring was hydroxylated, producing 2-hydroxyphenylacetic acid with 51 % yield isolated. In addition, the 4 fluorophenylacetonitrile was exclusively biotransformed to 4-fluorophenylacetic acid by Aspergillus sydowii Ce19 (yield051 %). The enzymatic biotransformation of nitriles is not trivial, and here, we describe an effi cient method for production of phenylacetic acids in mild conditions. [2]
Esterification Reaction
A preparation method for methyl 2‑hydroxyphenylacetate has been publicly reported, comprising the following steps: In step S1, a solid catalyst is added to 2‑hydroxyphenylacetic acid, followed by heating under reflux, during which 2-Hydroxyphenylacetic acid undergoes dehydration to form benzofuran‑2‑one. In step S2, methanol is introduced into the reaction system from step S1 to carry out a transesterification reaction, yielding methyl 2‑hydroxyphenylacetate. In step S3, the catalyst is filtered off for repeated reuse. The resulting solution is concentrated and cooled to crystallize, affording methyl 2‑hydroxyphenylacetate. The method provided by this invention is simple and easy to operate, with high conversion efficiency. The solid catalyst can be separated and reused multiple times without significant loss of catalytic activity. [3]
Chemical Applications
2-Hydroxyphenylacetic acid is a versatile and important fine chemical intermediate, also known as o‑hydroxyphenylacetic acid or 2‑(2‑hydroxyphenyl)acetic acid. With the chemical formula C₈H₈O₃, 2-Hydroxyphenylacetic acid typically appears as a beige to grayish‑yellow powder and exhibits certain solubility in water. The structure of 2-Hydroxyphenylacetic acid contains hydroxyl, carbonyl, and methylene groups, all of which are chemically reactive, and it can undergo self‑condensation and cyclization reactions. Due to its pronounced biological activity, 2‑hydroxyphenylacetic acid is widely employed in the synthesis of pesticides and pharmaceuticals. Its primary application lies in the production of methoxyacrylate‑type fungicides, such as azoxystrobin, and their corresponding active intermediates. [4]
References
[1] Yan, J. T. Preparation method of 2?hydroxyphenylacetic acid: CN 201310099985 [P].
[2] Oliveira, J. R.; Mizuno, C. M.; Seleghim, M. H. R.; et al. Biotransformation of phenylacetonitrile to 2?hydroxyphenylacetic acid by marine fungi. Marine Biotechnology 2013, 15, 97–103.
[3] Cao, J. L. Preparation method of methyl 2?hydroxyphenylacetate: CN 201610744897.8 [P].
[4] Yang, G. Z.; Guo, T. T.; Dong, P.; et al. Applications of 2?hydroxyphenylacetic acid in organic synthesis. Today's Agrochemicals 2017,3, 2.
You may like
Lastest Price from 2-Hydroxyphenylacetic acid manufacturers

US $30.00/kg2025-04-21
- CAS:
- 614-75-5
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 100 tons

US $1.00/kg2025-04-21
- CAS:
- 614-75-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt