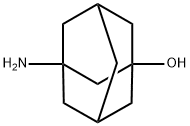
Synthesis and Medical Application of 3-Amino-1-Hydroxyadamantane
3-Amino-1-Hydroxyadamantane is an important 1,3-disubstituted adamantane amino alcohol derivative, which exists as a white to yellowish solid powder under standard temperature and pressure. Characterized by high polarity and excellent chemical stability, this compound is insoluble in water but readily soluble in organic solvents such as ethyl acetate and alcohols. In the field of chemical synthesis, 3-Amino-1-Hydroxyadamantane serves primarily as a key synthetic intermediate for the pharmaceutical molecule Vildagliptin—an oral anti-diabetic drug belonging to the dipeptidyl peptidase-4 (DPP-4) inhibitor class, developed by Novartis in Switzerland. Vildagliptin was first synthesized in 1998 by Dr. Edwin Villhauer and received approval for marketing in the European Union in 2008.
Synthesis
A patent has reported a method for preparing 3-amino-1-hydroxyadamantane, which comprises the following steps: adding adamantanamine hydrochloride in batches into a nitrating agent, reacting in an ice-water bath for 1–2 hours, and then continuing the reaction at room temperature for 1–30 hours to obtain a pale-yellow liquid; pouring the resulting pale-yellow liquid into ice, stirring continuously for 0.5–2 hours to obtain a bluish-green liquid; while stirring, adding a solid alkali into the solution from step (2) while maintaining the temperature below 80°C, adjusting the pH to 10–12, stirring for 30 minutes, followed by suction filtration; extracting the reaction mixture with dichloromethane, drying over anhydrous sodium sulfate, evaporating the dichloromethane, and recrystallizing with ethyl acetate to yield a white solid. As a key intermediate, 3-amino-1-hydroxyadamantane is synthesized via this process, which offers advantages such as readily available starting materials, simple operation, short synthetic route, and environmental friendliness, with a product yield reaching 75%. This method is suitable for industrial-scale production of 3-Amino-1-Hydroxyadamantane, and also reduces the synthesis cost of the diabetes therapeutic drug Vildagliptin, demonstrating promising application prospects. [1]
Acylation reaction
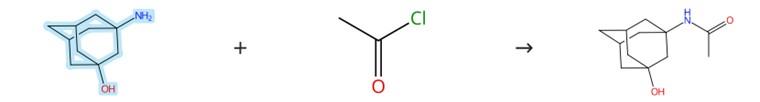
Figure1: Acylation reaction of 3-Amino-1-Hydroxyadamantane
To the pre-dissolved mixture of 3-Amino-1-Hydroxyadamantane, DIPEA, and THF maintained at 0°C, acetyl chloride was added dropwise, after which the reaction mixture was stirred at room temperature for 3 hours. Upon completion of the reaction, ice was introduced into the mixture to quench the process. The organic compounds were then extracted using ethyl acetate (three times, each with 50 mL), and the combined organic extracts were subsequently washed with brine. After drying over anhydrous sodium sulfate, the volatiles were evaporated to afford the crude residue. To this residue, isopropyl acetate (20 mL) was added, and the mixture was heated to achieve complete dissolution. Finally, the solution was cooled to allow crystallization, yielding the derivative in which the amino unit of the 3-Amino-1-Hydroxyadamantane structure had been acetylated. [2]
Medical Application
Starting with (S)-pyrrolidine-2-carbonitrile p-toluenesulfonate (W1) and chloroacetyl chloride as raw materials, the key intermediate (S)-1-(2-chloroacetyl)pyrrolidine-2-carbonitrile (W2) is synthesized through chloroacetylation. Subsequently, W2 undergoes nucleophilic substitution reaction with 3-Amino-1-Hydroxyadamantane to produce the active pharmaceutical ingredient Vildagliptin. Single-factor experiments were used to optimize the two-step process. The results show that the optimal reaction conditions are as follows: the molar ratio of W1 to chloroacetyl chloride and triethylamine is 1:1.1:1.2, and the reaction is carried out at 5°C for approximately 2 hours to obtain W2; the molar ratio of W2 to 3-Amino-1-Hydroxyadamantane is 1:2.0, and the reaction proceeds at 45°C for 12 hours to yield Vildagliptin. As a critical reactant in this synthesis, 3-Amino-1-Hydroxyadamantane enables efficient production under these optimized conditions. The final product was confirmed by infrared spectroscopy for its chemical structure, and its quality was determined via high-performance liquid chromatography, with purity reaching 99.9% and an overall yield of 76.5%. This preparation process is simple to operate, proceeds under mild reaction conditions, and achieves high purity and yield, making it suitable for industrial production. [3]
Reference
[1] ZHAO S Y,. A preparation method of 3-amino-1-adamantanol: CN 201010114206[P].
[2] Chandrasekhar, Manchikanti M.; et al, Synthesis, infra red and nmr intensities of amphiphilic adamantane derivatives: Impact of hydroxylation, acetylation and chlorination, Journal of Molecular Structure 2024, 1318, 139265.
[3] Wang L X, Liu X Y, Wang P, Wang Y D, Liang B C. Study on the synthesis process of vildagliptin[J]. Coal and Chemical Industry, 2022, 45: 129-132.
You may like
See also
Lastest Price from 3-Amino-1-adamantanol manufacturers

US $5.00-0.50/KG2025-05-30
- CAS:
- 702-82-9
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS

US $99.00-66.00/kg2025-04-21
- CAS:
- 702-82-9
- Min. Order:
- 0.0010000000474974513kg
- Purity:
- 99%
- Supply Ability:
- 5000