Synthesis and Metabolism of N,N-Diethylformamide
N,N-Diethylformamide is a polar organic solvent with excellent solvency and is miscible with most organic solvents. N,N-Diethylformamide can be synthesized via the carbonylation of diethylamine in methanol solvent using sodium methoxide as a catalyst under heating and high pressure. This substance is primarily used as a polar organic solvent and pharmaceutical intermediate, with applications in polyurethane resins, electronic circuit board cleaning, and as a solvent in lithium-ion battery slurries.
Synthesis
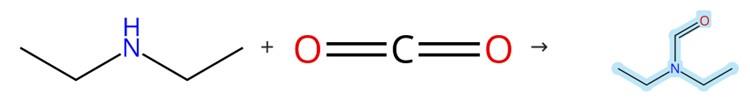
Figure1: Synthesis of N,N-Diethylformamide
The formylation reaction was conducted in a 15 mL stainless steel autoclave, which was sequentially charged at room temperature with [TMA][2‑Tp] (0.05 mmol/mL in MeCN), N-methylaniline (1.0 mmol), phenylsilane (1.0 mmol), and CH₃CN (0.8 mL). The reactor was pre‑heated to 80 °C for 5 minutes, after which 1.0 MPa of CO₂ was introduced, and the autoclave was maintained at this temperature for the reaction. Following the reaction, the autoclave was cooled in an ice‑water bath, and the excess CO₂ was carefully vented. The resulting solution was transferred into a 10 mL volumetric flask and diluted to volume with MeCN. The yield was determined by GC external standard analysis, and each test was repeated at least three times, with the average value recorded. For product isolation, the residue was purified by column chromatography using ethyl acetate–petroleum ether as the eluent to give N,N-Diethylformamide. [1]
Metabolism
N,N-Dimethylformamide (DMF) and N,N-Diethylformamide (DEF) are two hepatotoxic solvents whose metabolic pathways in humans have not yet been elucidated, prompting an investigation into the specific P450 isoforms responsible for their microsomal oxidation through the utilization of twelve distinct human liver samples, a panel of human recombinant P450 isoforms including 1A1, 1A2, 2B6, 2C10, 2E1, and 3A4, as well as chemical and immunological inhibition techniques. When correlation analyses were conducted using enzymatic markers within human liver microsomes, the hydroxylation rate of p-nitrophenol demonstrated a significant correlation (r=0.87) with the dealkylation rate of DMF; however, N,N-Diethylformamide did not exhibit a similar correlation with this enzymatic marker, indicating distinct metabolic profiles between the two solvents. Among the various recombinant P450 enzymes evaluated, only isoform 2E1 was capable of oxidizing DMF, whereas N,N-Diethylformamide was found to be oxidized by multiple isoforms including 2E1, 2C10, and 3A4, highlighting a broader range of enzymatic involvement in its metabolism. The application of 4-methylpyrazole and anti-human 2E1 IgG resulted in strong inhibition of DMF demethylation, but these inhibitors only partially suppressed the deethylation of N,N-Diethylformamide, further confirming that while 2E1 plays a dominant role in DMF metabolism, N,N-Diethylformamide is metabolized through a more complex pathway involving several P450 enzymes. These collective findings indicate that the role of 2E1 is crucial in DMF metabolism, and its expression level may represent an important determinant of human susceptibility to the hepatotoxic effects of this particular solvent. [2]
Oxidized by P450 system
It has been demonstrated that N,N-Dimethylformamide (DMF) and N,N-Diethylformamide (DEF) are oxidized by the P450 system on their alkyl moiety, yielding hydroxymethyl-methylformamide (HMMF) and hydroxyethyl-ethylformamide (HEEF), respectively. These intermediates then undergo chemical or enzymatic decomposition to form N-methylformamide (NMF) and N-ethylformamide (MEF), which are further metabolized via P450-mediated oxidation to produce reactive carbamoylating intermediates—methylisocyanate (MIC) from DMF and ethylisocyanate (EIC) from DEF. These metabolites subsequently react with water, nucleophilic cellular macromolecules, or glutathione, leading to the formation of thiocarbamates and ultimately resulting in the urinary excretion of N-acetyl-S-N-methyl-carbamoyl-cysteine (AMCC) or N-acetyl-S-N-ethyl-carbamoyl-cysteine (AECC). In rodents, the P450 2E1 isoform has been identified as an effective catalyst in the biotransformation of DMF and NMF, and recent findings have also demonstrated its crucial role in the oxidation of DEF. However, information on the in vitro metabolism of DMF in humans remains limited, and no data are currently available regarding DEF. To address this gap, the present study was undertaken to investigate the oxidative metabolism of both DMF and DEF using liver microsomes derived from 12 human donors, as well as human recombinant P450 enzymes expressed in E. coli, with particular attention to the role of N,N-Diethylformamide as a substrate for human P450-mediated oxidation. [2]
Reference
[1] Wu, Jiakai; et al, Highly Efficient Thiolate-Based Ionic Liquid Catalysts for Reduction of CO2: Selective N-Functionalization of Amines to Form N-Formamides and N-Methylamines, Chemistry - A European Journal (2024), 30(32), e202304315
[2] Amato G, Grasso E, Longo V, et al. Oxidation of N, N-dimethylformamide and N, N-diethylformamide by human liver microsomes and human recombinant P450s[J]. Toxicology letters, 2001, 124: 11-19.
You may like
Lastest Price from N,N-Diethylformamide manufacturers

US $79.00-38.00/kg2025-04-21
- CAS:
- 617-84-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton

US $0.00-0.00/KG2025-04-15
- CAS:
- 617-84-5
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg