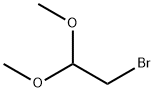
Synthesis and Nucleophilic Substitution Reaction of Bromoacetaldehyde dimethyl acetal
Bromoacetaldehyde dimethyl acetal is an alkyl bromide that appears as a colorless to pale orange liquid under ambient temperature and pressure. It exhibits relatively high density and poor chemical stability, remaining stable toward alkaline substances but being prone to hydrolysis and degradation upon exposure to acidic conditions. Primarily used as an organic synthesis intermediate and a fundamental chemical raw material for pharmaceutical molecules, Bromoacetaldehyde dimethyl acetal has been reported in studies to be applicable in the synthesis of 2-acylthiazoles via cyclocondensation with α-oxothioamides.
Activation Method
A research study has reported a method for activating 2-bromo-1,1-dimethoxyethane. The procedure involves sequentially adding an appropriate amount of bromine and vinyl acetate into a reaction vessel and mixing the solution. The mixture is stirred at 55°C, followed by the addition of a certain proportion of an alcohol-water mixture for alkoxylation. The reaction mixture is then heated and distilled. After distillation, the product is filtered using a circulating filtration device, and silver bicarbonate powder is added. The mixture is washed with an aqueous sodium hydroxide solution. The washed sample is separated and dried to obtain the product Bromoacetaldehyde dimethyl acetal. Subsequently, an additional 0.1 g to 0.5 g of silver bicarbonate powder is introduced, yielding the activated and decomposition-resistant final product 2-bromo-1,1-dimethoxyethane. In this invention, the addition of silver bicarbonate powder removes excess bromide ions, thereby enhancing the stability and activity of Bromoacetaldehyde dimethyl acetal, making it less prone to decomposition. [1]
Synthesis
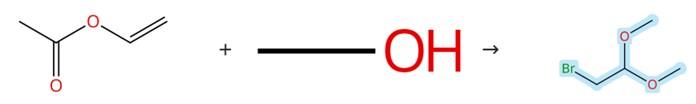
Figure1: Synthesis of Bromoacetaldehyde dimethyl acetal
In a 500 mL three-neck round-bottom flask equipped with a magnetic stirring bar, two dropping funnels, and a thermometer, methanol (140 mL) was charged. The flask was cooled in an ice-cold bath (or a NaCl/ice bath), and then bromine (Br₂, 102.98 g, 0.64 mol) and vinyl acetate (55.52 g, 0.64 mol) were added simultaneously via the dropping funnels under vigorous stirring, while carefully controlling the addition rate to ensure the internal temperature did not exceed 5 °C. After complete addition, the resulting colorless solution was allowed to stand overnight at room temperature. The flask was subsequently cooled to approximately –10 °C, and the reaction mixture was carefully poured into a 1 L glass vessel that had been pre-cooled in an ice-cold bath and charged with ice-cold water (80 mL) and sodium bicarbonate (NaHCO₃, 56.10 g, 0.67 mol) under vigorous magnetic stirring. Dichloromethane (CH₂Cl₂, 80 mL) was then added, and the mixture was stirred for 30 minutes under continued ice-bath cooling until foaming subsided completely. The organic layer was separated, and the aqueous layer was further extracted with dichloromethane (10 mL followed by 30 mL). The combined organic phases were washed with brine (20 mL), dried over anhydrous sodium sulfate (Na₂SO₄) for 30 minutes, and filtered. Dichloromethane was removed under reduced pressure, and the product Bromoacetaldehyde dimethyl acetal was collected by distillation at 70–82 °C under 80 mm Hg vacuum. [2]
Nucleophilic Substitution Reaction
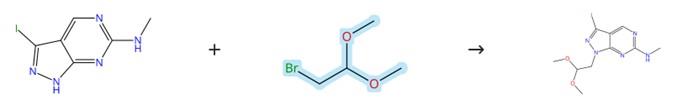
Figure2: Nucleophilic Substitution Reaction of Bromoacetaldehyde dimethyl acetal
A suspension of 3-iodo-N-methyl-1H-pyrazolo[3,4-d]pyrimidin-6-amine (7) (0.182–0.461 mmol) in DMF (3.5 mL) was prepared. To this suspension, sodium hydride (1.4 equiv., 60% dispersion in mineral oil) was added, and the mixture was stirred until gas evolution ceased. Subsequently, Bromoacetaldehyde dimethyl acetal (1.5 equiv.) was introduced into the reaction mixture. The mixture was then heated at 150°C under microwave irradiation for 85 minutes. After completion of the reaction, the mixture was partitioned between ethyl acetate and water. The organic layer was separated, washed with brine, and dried over anhydrous magnesium sulfate. The solvent was removed under reduced pressure, and the crude product was purified by flash chromatography on silica gel using a gradient of ethyl acetate in hexane (0–40%) to afford 1-(2,2-dimethoxyethyl)-3-iodo-N-methyl-1H-pyrazolo[3,4-d]pyrimidin-6-amine. [3]
Reference
[1] Li, B.; Zhou, R. J.; Chen, S. H.; et al. A method for activating 2-bromo-1,1-dimethoxyethane: CN201810751991.5[P].
[2] Fesenko, Anastasia A.; et al, Synthesis of functionalized tetrahydro-1,3-diazepin-2-ones and 1-carbamoyl-1H-pyrroles via ring expansion and ring expansion/ring contraction of tetrahydropyrimidines, Organic & Biomolecular Chemistry 2012, 1, 447-462.
[3] Ayala-Aguilera, Cecilia C.; et al, Ligand-centred phenotype-driven development of potent kinase inhibitors against oesophageal cancer, C Medicinal Chemistry 2025, 16, 379-391.
You may like
Related articles And Qustion
See also
Lastest Price from Bromoacetaldehyde dimethyl acetal manufacturers

US $0.00-0.00/KG2025-05-16
- CAS:
- 7252-83-7
- Min. Order:
- 1KG
- Purity:
- 99.0%
- Supply Ability:
- 10000KGS

US $0.00/Kg/Drum2025-04-21
- CAS:
- 7252-83-7
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 1000KGS