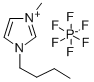
Synthesis and Thermodynamic Properties of 1-Butyl-3-methylimidazolium hexafluorophosphate
1-Butyl-3-methylimidazolium hexafluorophosphate is an organic phosphate compound used as an ionic liquid. It possesses unique solvation and extraction properties, making it useful for extracting complex molecules from natural products and for various applications in medicinal chemistry research. As a room-temperature ionic liquid, 1-Butyl-3-methylimidazolium hexafluorophosphate is easy to synthesize and purify, has a melting point of -8°C, and exhibits solvent behavior with considerable polarity comparable to that of ethanol.

Figure1: Picture of 1-Butyl-3-methylimidazolium hexafluorophosphate
Background Introduction
Room-temperature ionic liquids (RTILs) have attracted significant attention due to their potential applications in the chemical industry as environmentally friendly and highly selective solvents for chemical reactions, homogeneous catalysis, extraction separations, and electrochemical processes. These liquids can effectively reduce the costs associated with volatile organic solvent losses and environmental pollution. Investigating the thermodynamic properties of RTILs contributes to a deeper understanding of their structural characteristics and provides theoretical support for the development of synthesis processes and application technologies. However, to date, no RTIL has been comprehensively and systematically studied in terms of its thermodynamic properties. When conducting physical property measurements on any RTIL sample, determining its purity is essential. In this study, the purity of the 1-butyl-3-methylimidazolium hexafluorophosphate test sample was determined using the fractional melting method. Additionally, the heat capacity of 1-butyl-3-methylimidazolium hexafluorophosphate in the condensed state was systematically reported via adiabatic calorimetry (5–300 K) and scanning calorimetry (300–500 K). Thermodynamic parameters related to fusion and glass transition were also provided. The study found that the temperature dependence of the heat capacity of 1-butyl-3-methylimidazolium hexafluorophosphate significantly differs from that of conventional molecular liquids and ionic crystals, with the most notable difference being an abnormally high specific heat capacity below 50 K. This anomaly may arise from an unusually low characteristic temperature of the 1-butyl-3-methylimidazolium hexafluorophosphate crystal or from a significant electronic contribution to the heat capacity in this temperature range. [1]
Thermodynamic Properties
Thermodynamic functions for 1-Butyl-3-methylimidazolium hexafluorophosphate are reported in a range of temperatures from (5 to 550) K, based on new measurements by calorimetry. Heat capacities of the crystal, glass, and liquid phases for 1-Butyl-3-methylimidazolium hexafluorophosphate were measured with a pair of calorimeters. A vacuum-jacketed adiabatic calorimeter was used at temperatures between (5 and 310) K, and a heat bridge-scanning calorimeter was used from (300 to 550) K. With the adiabatic calorimeter, the fusion Tfus ) 283.51 K, ¢cr 1 H°m ) 19.60 kJâmol-1, and the glass transition Tg ) 190.6 K were observed. The 1-Butyl-3-methylimidazolium hexafluorophosphate test sample was determined to have a mole fraction purity of 0.9956 by a fractional melting analysis. Densities of the liquid were measured in a range of temperatures from (298 to 353) K with a pycnometer equipped with a capillary neck. An unexpected endothermal transition, with a very small enthalpy change of 0.25 Jâg-1 (0.071 kJâmol-1), was observed in a range of temperatures from (394 to 412) K. [1]
Synthesis
1-Butyl-3-methylimidazolium chloride was synthesized by reacting 1-methylimidazole with chlorobutane . Typically, 100mL of 1-methylimidazole (103g, 1.25mol) was mixed in a 1-L round-bottom flask with 140mL of 1-chloro butane (124g, 1.34mol). A stirrer bar was added and the mixture was heated under reflux at 70°C for 72h. A viscous pale yellow liquid was obtained and it was washed twice with 100mL ethyl acetate. After drying overnight at 100 °C, the pale yellow, highly vis cous BMIM-Cl (208g, 1.19mol) was obtained in 95% yield. It should be pointed out that BMIM-Cl is not as thermally sta ble as other ionic liquids. 1-Butyl-3-methylimidazolium hexafluorophosphate and BMIM bis(triflate)amide were reported to be stable at 300 and 400 °C. Upon drying BMIM-Cl, we observed that at temperatures as low as 120°C, the pale yellow color turned amber. We increased the temperature to 140°C and observed emission of basic fumes and further darkening of the liquid phase. BMIM-Cl seems to boil but, exposed to the atmosphere, it actually decomposes at or above 150°C. 1-Butyl-3-methylimidazolium hexafluorophosphate was obtained by metathesis of the chloride ion of BMIM-Cl using hexafluorophosphoric acid. [2]
Reference
[1] Kabo G J, Blokhin A V, Paulechka Y U, et al. Thermodynamic properties of 1-butyl-3-methylimidazolium hexafluorophosphate in the condensed state[J]. Journal of Chemical & Engineering Data, 2004, 49: 453-461.
[2] Carda–Broch S, Berthod A, Armstrong D W. Solvent properties of the 1-butyl-3-methylimidazolium hexafluorophosphate ionic liquid[J]. Analytical and bioanalytical chemistry, 2003, 375: 191-199.
You may like
Lastest Price from 1-Butyl-3-methylimidazolium hexafluorophosphate manufacturers

US $300.00/KG2026-03-30
- CAS:
- 174501-64-5
- Min. Order:
- 1KG
- Purity:
- 98%
- Supply Ability:
- UP TO TONS

US $0.00/KG2025-04-21
- CAS:
- 174501-64-5
- Min. Order:
- 25KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month