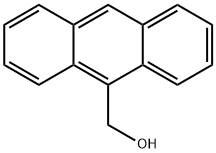
Synthesis and transformations of 9-Anthracenemethanol
9-Anthracenemethanol is an aryl benzyl alcohol compound that exists as a yellow crystalline solid at ambient conditions, exhibiting notable fluorescence and excellent chemical stability. It is insoluble in water but soluble in strongly polar alcoholic organic solvents. In organic synthesis, 9-Anthracenemethanol serves primarily as a starting material for the preparation of organic luminescent materials and can be employed in the synthesis of organic dyes with extended conjugated systems.
Synthesis

Figure1: Synthesis of 9-Anthracenemethanol
A solution of 9-Anthracenemethanol (2 g, 9.7 mmol) in tetrahydrofuran (15 mL) was stirred at room temperature. To this mixture, a solution of sodium borohydride (0.35 g, 10.04 mmol) in water (3 mL) and THF (1 mL) was added dropwise over approximately 3 minutes. After stirring for 1 hour, the reaction mixture was poured into ice‑cold water (50 mL), causing 9‑anthracenemethanol to precipitate as pale white crystals with a yield of 97%. [1]
Vibrational and electronic spectra
9-Anthracenemethanol’s equilibrium geometry, FT‑IR, and UV–vis spectra were calculated using both RHF and DFT methods with 6‑311G** and LANL2DZ basis sets. The DFT approach yielded more reasonable results than RHF, reproducing the observed vibrational frequencies with good accuracy and assigning all fundamental modes, including C–H, C–O, O–H stretching and ring vibrations. Electronic absorption spectra of 9-Anthracenemethanol were predicted via TDDFT in the gas phase and PCM‑TDDFT in ethanol (CH₃CH₂OH) and acetonitrile (CH₃CN) solutions. Experimentally, the strongest absorption band in solution appears at 244.5 nm in ethanol and 243.6 nm in acetonitrile, while the corresponding TDDFT predictions give peaks at 248.6 nm and 247.5 nm, respectively. The close agreement between the theoretical and experimental maxima confirms the reliability of the computational models. [2]
Diels-Alder reaction
The Diels–Alder reaction between 9-Anthracenemethanol and dimethyl acetylenedicarboxylate initially generates an adduct in which the hydroxyl group of the 9‑substituent condenses with a neighboring carboxylate moiety to form a lactone derivative. This lactone can be opened by treatment with an alcohol to afford the corresponding Diels–Alder adduct. When the alcohol employed for lactone opening differs from the alkoxy group originally present in the diester reagent, the process yields a regiospecific adduct wherein the newly introduced alkoxy group is located ortho to the 9‑substituent. A mixture of 9-Anthracenemethanol (1.7 g, 8.16 mmole) and dimethyl-acetylene dicarboxylate (DMAD) 8 (1.5 mL, 12.2 mmole) in 10 mL toluene was refluxed for 24 hr. After evaporating the solvent under reduced pressure the mixture was chromato graphed on a column of silica gel using ethyl acetate and hexane (1:4 v/v) as the eluent. The product obtained 9a was found to be a lactone derivative. Yield: 85%; m.p. 174-175oC. [2]
Acylation Reaction
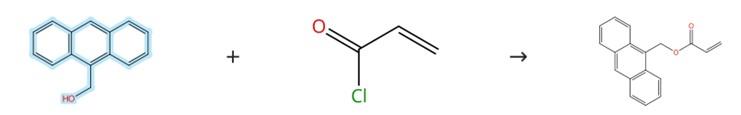
Figure2: Acylation Reaction of 9-Anthracenemethanol
Under a nitrogen atmosphere, a solution containing 1.250 g (6 mmol) of 9-Anthracenemethanol and 0.073 g (0.6 mmol) of DMAP in 250 mL of super‑dry dichloromethane, along with 1.668 mL (12 mmol) of triethylamine, was prepared in an ice bath. To this mixture, a solution of 0.975 mL (12 mmol) of acryloyl chloride in 15 mL of dichloromethane was added dropwise. After stirring at room temperature for 4 hours, the reaction mixture was diluted with additional dichloromethane and washed sequentially with 0.02 M hydrochloric acid and saturated sodium hydrogen carbonate solution. The organic phase was then concentrated using a rotary evaporator to afford the product. [3]
Condensation Reaction
In a round‑bottom flask, 9-Anthracenemethanol (3.00 g), 1‑(3‑dimethylaminopropyl)‑3‑ethylcarbodiimide hydrochloride (3.31 g, 17.3 mmol, 1.2 equiv), and 4‑dimethylaminopyridine (0.211 g, 1.73 mmol, 0.12 equiv) were combined. The mixture was purged with argon, and dichloromethane (60 mL) was added, followed by stirring at room temperature until a homogeneous solution was obtained. 3‑Cyclopentene‑1‑carboxylic acid (1.94 g) was then introduced slowly dropwise. After stirring for 24 hours at room temperature, the reaction mixture was concentrated to give the crude product, which was purified by column chromatography using a 50% hexane/dichloromethane mixture as the eluent. The collected fractions were evaporated on a rotary evaporator, and the resulting solid was dried overnight in a vacuum oven. [4]
Reference
[1] Singh M D, Ningombam A. Diels-Alder reaction of 9-anthracenemethanol and dimethylacetylene-dicarboxy-late; potential route for the synthesis of regiospecific products of 9-substituted anthracene with unsymmetrical[J]. Indian J. Chem, 2010, 49: 77-83.
[2] Kou S, Zhou H, Tang G, et al. Experimental and DFT studies on the vibrational and electronic spectra of 9-anthracenemethanol[J]. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2012, 96: 768-775.
[3] Shen, Ying; et al, Spatiotemporal Regulation Enabling Photo-Dimerizable Gel Networks Toward Multi-Channel Information Encryption, Advanced Functional Materials 2026, 36, e13532.
[4] Lee, Juho ; et al, High-Performance Dynamic Photo-Responsive Polymers With Superior Closed-Loop Recyclability, Advanced Functional Materials (2025), 35(8), 2414842.
You may like
Lastest Price from 9-Anthracenemethanol manufacturers

US $0.00/KG2025-04-21
- CAS:
- 1468-95-7
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month
US $0.00-0.00/kg2025-04-21
- CAS:
- 1468-95-7
- Min. Order:
- 1kg
- Purity:
- 98%+
- Supply Ability:
- 10000kgs per Month