Synthesis Methods and Biological Activity of L-Selenomethionine
L-Selenomethionine is the L-enantiomer of selenomethionine. It is a selenium analog of methionine and occurs naturally in plants such as cabbage and garlic, as well as in some microorganisms. As one of the important natural organic selenium sources, L-Selenomethionine exhibits good bioavailability and is commonly used as a nutritional selenium supplement. Selenium is an essential trace element for the human body, functioning as a cofactor for glutathione peroxidase and exerting antioxidant effects. [1]

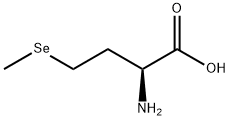
Figure1: Picture of L-Selenomethionine
Synthesis Methods
The preparation methods for L-Selenomethionine mainly include chemical synthesis, biosynthesis, and structural modification using L-Selenomethionine as the starting material. Chemical synthesis is the primary approach for producing L-Selenomethionine in both laboratory and industrial settings. According to literature reviews, the main methods include the 2-amino-4-bromobutyric acid hydrobromide substitution method, the 2-amino-4-benzylselenomethionine substitution method, the three-pot boiling method, the Na/LiqNH₃ method, the acrolein method, the butyrolactone method, and the methionine method. Among these, the methionine method is more commonly used in industrial production due to its process advantages. L-Selenomethionine also exists in nature and can be obtained through biosynthetic pathways. Plants (such as cabbage and garlic), yeasts (such as Hansenula polymorpha), and certain bacteria are capable of absorbing inorganic selenium (such as selenite or selenate) from soil or culture media and converting it into selenomethionine. Through optimization of fermentation processes, such as using fructose as a carbon source, the content of selenomethionine in yeast can be increased. Using L-Selenomethionine as a raw material, derivatives with specific functions can be synthesized through chemical modification. For example, L-Selenomethionine can undergo Schotten-Baumann condensation reactions with acyl chlorides (such as lauroyl chloride and oleoyl chloride), followed by methyl esterification, to produce N-acyl-L-selenomethionine methyl ester derivatives.
Biological Activity
As a trace element essential for vertebrates and involved in a series of vital metabolic functions, selenium is provided in the form of L-selenomethionine, and considering the purity of the L-selenomethionine under application along with the established metabolic pathways of selenomethionine, the FEEDAP Panel considers L-selenomethionine to be safe for all animal species provided that the maximum total selenium level authorized in feed is respected; L-selenomethionine is expected to result in an increase in selenium deposition in animal tissues and products that is similar to that resulting from other sources of selenomethionine. To ensure consumer safety with respect to food originating from animals fed L-selenomethionine, the FEEDAP Panel concludes that dietary selenium supplementation from the additive should not exceed a maximum of 0.2 mg Se per kg of complete feed, and in the absence of specific data, the additive should be considered an irritant to skin and eyes, a skin sensitiser, and potentially harmful by inhalation. L-selenomethionine is considered by the FEEDAP Panel to pose no additional risk to the environment compared with other selenium sources it would replace, as long as the maximum authorized content in feedingstuffs is not exceeded, and it is recognized as an efficient source of selenium for all species, a conclusion derived from studies with laying hens and pigs for fattening as well as from literature describing the microbial incorporation of selenium from organic sources in the rumen for ruminants. The FEEDAP Panel also made several recommendations concerning the specification of L-selenomethionine, its use in premixtures, its use in water for drinking, and risk reduction measures when handling the additive. [1-2]
Industrial Applications
L-Selenomethionine is an important organic selenium source. As a significant natural food-form selenium, it is considered a safe, effective, and highly bioavailable form of selenium. In the feed industry, zinc-L-selenomethionine preparations have been approved by the European Union as feed additives for all animal species, and zinc-L-selenomethionine has also been approved in the United States as a nutritional source of selenium in broiler chicken feed.
Biomedical research
In biomedical research, L-Selenomethionine is a research target classified as a human endogenous metabolite. Studies have shown that selenium may have chemopreventive effects against prostate cancer. As a selenium analog of methionine, L-Selenomethionine can act as a reactive oxygen species scavenger and can be oxidized by peroxynitrite to methionine selenoxide, which can subsequently be reduced in the presence of glutathione. L-Selenomethionine exhibits cytotoxicity against various cancer cells, including those of lung cancer, breast cancer, colorectal cancer, prostate cancer, and melanoma. In a mouse model of Alzheimer's disease, L-Selenomethionine promotes hippocampal neurogenesis through the PI3K-Akt-GSK3β-Wnt pathway. Studies have also shown that L-Selenomethionine has protective effects against tissue damage induced by ammonia, methylmercury, and mycotoxins such as aflatoxin B1 and ochratoxin A. Through chemical modification of L-Selenomethionine, such as the synthesis of N-acyl derivatives, its application range in fields including food, medicine, and cosmetics can be expanded.
Reference
[1] EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). Scientific Opinion on the safety and efficacy of L‐selenomethionine as feed additive for all animal species[J]. EFSA Journal, 2013, 11(5): 3219.
[2] Schrauzer G N. Selenomethionine: a review of its nutritional significance, metabolism and toxicity[J]. The Journal of nutrition, 2000, 130(7): 1653-1656.
You may like
See also
Lastest Price from L-Selenomethionine manufacturers

US $0.00/kg2025-11-19
- CAS:
- 3211-76-5
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise

US $0.00-0.00/kg2025-04-18
- CAS:
- 3211-76-5
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 50kg/M

![13080-86-9 Preparation Method2,2-Bis[4-(4-aminophenoxy)phenyl]propane](/NewsImg/2026-03-22/6390981185175611622296370.jpg)