Synthesis of ML264
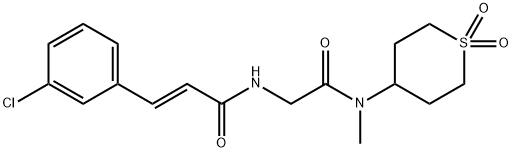
ML264, chemically known as (E)-3-(3-chlorophenyl)-N-(2-((1,1-dioxotetrahydro-2H-thiopyran-4-yl)(methyl)amino)-2-oxoethyl)acrylamide, is a third-generation small molecule compound derived from first-generation uHTS screening results. ML264 effectively inhibits KLF5 expression, reduces the proliferative capacity of colorectal cancer cell lines, and suppresses xenograft tumour growth in mouse models of primary tumour development. It can be synthesised through a three-step synthetic route.
Detailed experimental procedures for the preparation of ML264, (E)-3-(3-chlorophenyl)-N-(2-((1,1-dioxidotetrahydro-2H-thiopyran-4-yl)(methyl)amino)-2-oxoethyl)acrylamide.
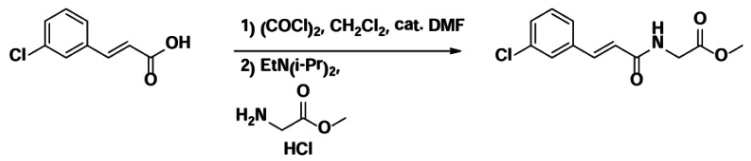
Step 1. Preparation of (E)-methyl 2-(3-(3-chlorophenyl)acrylamido)acetate
To (E)-3-(3-chlorophenyl)acrylic acid (1.0 g, 5.48 mmol) in dichloromethane (10 mL) was added anhydrous dimethylformamide (DMF) (0.08 mL, 1.0 mmol). This solution was cooled to 0°C and then oxalyl dichloride (0.57 mL, 6.53 mmol) was added dropwise. The reaction was allowed to warm to room temperature. After two hours at room temperature, the reaction mixture was re-cooled to 0°C and a mixture of the HCl salt of glycine methyl ester (1.38 g, 10.99 mmol) and diisopropyl ethyl amine (DIEA, 3.8 mL, 21.92 mmol) in dichloromethane (10 mL) was added slowly. The reaction was stirred at room temperature overnight. The solvent was removed in vacuo to obtain the crude product, which was purified by flash chromatography (AcOEt/Hex 10~100%) to obtain the title compound, 1.19g (86%).
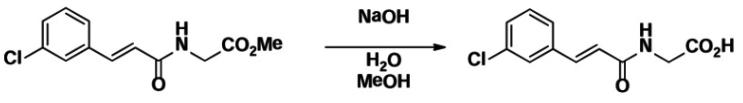
Step 2. Preparation of (E)-2-(3-(3-chlorophenyl)acrylamido)acetic acid
To a solution of (E)-methyl 2-(3-(3-chlorophenyl)acrylamido)acetate (1.553 g, 6.14 mmol) in MeOH (10 mL) was added 2.0 N NaOH (6.2 mL, 12.4 mmol). The mixture was stirred at room temperature for 1h, after which time analytical HPLC indicated that the reaction was complete. The mixture was acidified by with a 1N HCl solution. The solvent was removed in vacuo to obtain the crude which was used to the next step with no further purification.
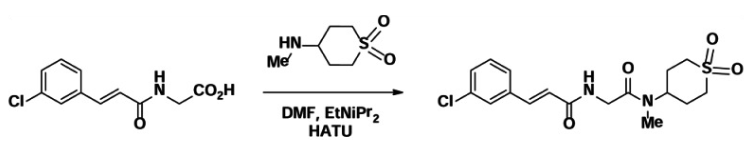
Step 3. Preparation of (E)-3-(3-chlorophenyl)-N-(2-((1,1-dioxidotetrahydro-2H-thiopyran-4-yl)(methyl)amino)-2-oxoethyl)acrylamide, ML264
To a mixture of (E)-2-(3-(3-chlorophenyl)acrylamido)acetic acid (31.5 mg, 0.13 mmol) in DMF (1 mL) was added DIEA (52 mg, 0.4 mmol) and HATU (50 mg, 0.13 mmol). The mixture was stirred for 5 min, and then 4-(methylamino)tetrahydro-2H-thiopyran 1,1-dioxide hydrochloride (26 mg, 0.13 mmol) was added. The reaction mixture was stirred at room temperature for 30 min. The completion reaction was monitored by analytical HPLC. The solvent was removed in vacuo to obtain the crude which was purified by prep-HPLC (Acetonitrile/MeOH(1:1)/water 40~100%) to obtain the title compound, 24.6 mg (49%).