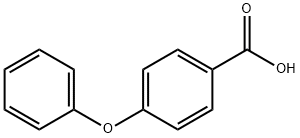
Synthetic Method and Amidation reaction of 4-Phenoxybenzoic acid
4-Phenoxybenzoic acid is a benzoic acid derivative that appears as a white to off-white solid powder under normal temperature and pressure, characterized by its significant acidity and excellent chemical stability. Although it is insoluble in water, it readily dissolves in alkaline aqueous solutions and alcohol-based organic solvents. 4-Phenoxybenzoic acid can be prepared from 4-phenoxyacetophenone through a selective oxidation reaction, and it is primarily utilized as an organic synthesis intermediate and as a raw material in pharmaceutical synthesis, with literature reports indicating its application in the preparation of the drug sitafloxacin.
Synthetic Method
Method 1
In this method, sodium hypochlorite is employed as the oxidizing agent in the presence of polyethylene glycol (PEG)-400 as a phase transfer catalyst to oxidize 4-phenoxyacetophenone, yielding 4-phenoxybenzoic acid. A systematic investigation was conducted to optimize the reaction conditions, including the molar ratio of starting materials, the selection and amount of phase transfer catalyst, and the pH of the reaction mixture. The optimal conditions were determined as follows: a molar ratio of 4-phenoxyacetophenone to sodium hypochlorite to PEG-400 of 1 mol : 4 mol : 150 mL, with the pH of the reaction mixture maintained between 12 and 13 (requiring the effective chlorine content of sodium hypochlorite to exceed 15%). Under these optimized conditions, the product yield of 4-Phenoxybenzoic acid exceeded 92%.
Method 2
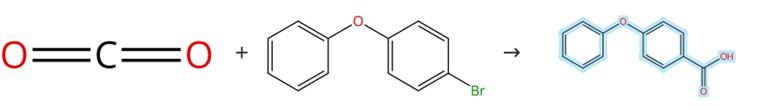
Figure1: Synthesis of 4-Phenoxybenzoic acid
Add Pd(dba)₂ (0.006 mmol), DPEPhos (0.0075 mmol), aryl bromide (0.3 mmol), and 1‑methyl‑2‑pyrrolidone (1 mL) to a 25 mL dried Schlenk tube, which is then subjected to vacuum for a short period. After introducing CO₂ (1 atm) into the reaction vessel, a solution of NEt₃ in 1‑methyl‑2‑pyrrolidone (0.6 M, 0.5 mL, 0.3 mmol) and a solution of PhSiH₃ in 1‑methyl‑2‑pyrrolidone (1.2 M, 0.5 mL, 0.6 mmol) are added via syringe, and the mixture is stirred at 60 °C in an oil bath for 18 h. Following cooling to room temperature, the reaction mixture is quenched with HCl (2 M) and extracted with ethyl acetate (10 mL × 3). The combined organic layers are washed with brine (10 mL), dried over anhydrous MgSO₄, and concentrated in vacuo, and the residue is purified by column chromatography on silica gel using 5:1 petroleum ether/ethyl acetate as the eluent to give 4-Phenoxybenzoic acid. [1]
Amidation reaction
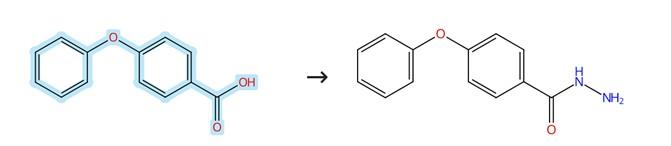
Figure2: Amidation reaction of 4-Phenoxybenzoic acid
To a solution of the 4-Phenoxybenzoic acid (1.0 mmol) in 10 mL of EtOH is added a catalytic equivalent of concentrated sulfuric acid (0.1 mmol), and the resulting mixture is refluxed for 8 hours. Hydrazine hydrate (85%, 5.0 mmol) is then slowly added, and the reaction mixture is refluxed for an additional 8 hours. After evaporation of the solvent in vacuo, the residue is diluted with EtOAc and H₂O, and the resulting mixture is extracted, followed by washing with brine. The organic layer is dried over Na₂SO₄, concentrated, and the crude product is purified by silica gel chromatography eluting with ethyl acetate/petroleum ether (1:15, v/v) to afford the desired product. [2]
Acyl chlorination
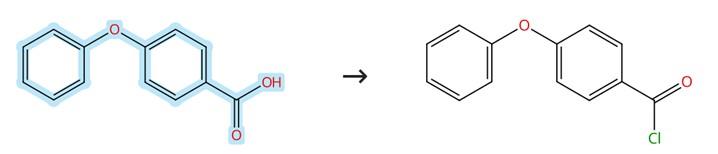
Figure3: Acyl chlorination of 4-Phenoxybenzoic acid
To a stirred solution of 4-Phenoxybenzoic acid (1 equivalent) in CH₂Cl₂ (0.5 M) containing a few drops of DMF at room temperature is added oxalyl chloride (1.05 equivalents) dropwise, and the resulting reaction mixture is stirred for 2 hours before being concentrated under reduced pressure, after which the residue is redissolved in an appropriate amount of anhydrous THF. [3]
Pharmaceutical Applications
4-Phenoxybenzoic acid has been shown to block the DNA binding activity of the human papillomavirus (HPV) E2 protein, thereby exhibiting potential relevance in antiviral research. In addition, 4-Phenoxybenzoic acid serves as a key intermediate in the synthesis of sitafloxacin, a fluoroquinolone antibiotic that holds promise for the treatment of Buruli ulcer, highlighting its importance in the development of antibacterial therapeutics.
Reference
[1] Li, Dan; et al, Palladium-Catalyzed Carbonylation of Aryl Bromides with Carbon Dioxide To Access Aryl Carboxylic Acids under Mild Conditions, Journal of Organic Chemistry 2023, 88, 5205-5211.
[2] Liang, Xuewu ; et al, Discovery of Novel RNA Demethylase FTO Inhibitors Featuring an Acylhydrazone Scaffold with Potent Antileukemia Activity, Journal of Medicinal Chemistry 2025, 68, 2742-2763.
[3] Han, Wenlai ; et al, Migrating Group Strategy for Remote Functionalization of Seven-Membered Rings, Journal of the American Chemical Society 2025, 147, 32077-32084.
You may like
See also
Lastest Price from 4-PHENOXYBENZOIC ACID manufacturers

US $10.00/KG2025-04-21
- CAS:
- 2215-77-2
- Min. Order:
- 100KG
- Purity:
- 99%
- Supply Ability:
- 100 mt

US $0.00-0.00/KG2025-03-03
- CAS:
- 2215-77-2
- Min. Order:
- 1KG
- Purity:
- 98.0%
- Supply Ability:
- 10000KGS