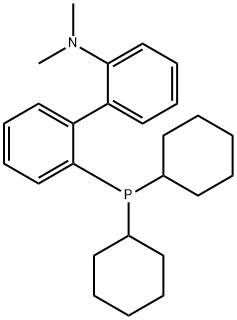
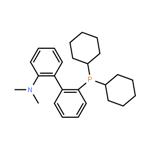
Synthetic Method and Catalytic Applications of 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl
2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl is an nitrogen-phosphine ligand that appears as a white to off-white solid powder under ambient conditions. It exhibits moderate basicity and good chemical stability. Insoluble in water, it is soluble in chloroform and ethyl acetate. 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl is relatively sensitive to oxidants; exposure to strong oxidizing agents can lead to degradation due to oxidation of the phosphorus atom within its structure. In organic synthesis, it is primarily employed as an organophosphine ligand capable of coordinating with various transition metal ions, and it holds certain applications in fundamental research within the field of organic synthetic methodology.
Synthetic Method
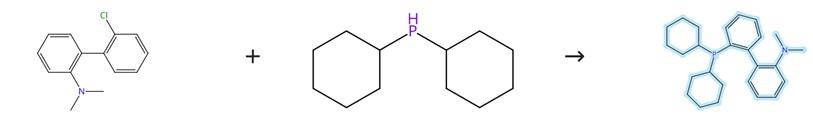
Figure1: Synthetic Method of 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl
An oven-dried 100 mL round-bottom flask was charged with aryl bromides (15.0 mmol) and 20 mL of THF. The resulting mixture was added dropwise at room temperature to a separate oven-dried 100 mL round-bottom flask containing magnesium turnings (0.40 g, 16.5 mmol) and a small amount of I₂. The mixture was cooled with an ice bath until the reaction was complete, after which a solution of diethyl phosphite (0.69 g, 5.0 mmol) in 2.0 mL of THF was added. The mixture was allowed to warm to room temperature and stirred for 8 h. Then, 5.0 mL of 3% HCl was added dropwise over 5 minutes with cooling in an ice bath, followed by the addition of 5.0 mL of MTBE and stirring for an additional 5 minutes. The upper organic phase was decanted from the formed gel. To the remaining gel, 30 mL of CH₂Cl₂ was added, and the mixture was agitated well for another 5 minutes. The resultant mixture was filtered through a fritted funnel packed with Celite. After washing the Celite with CH₂Cl₂ (2 × 30 mL), the combined organic phases were dried over MgSO₄, and the solvent was removed in vacuo. The residue was purified by flash column chromatography on silica gel using petroleum ether/ethyl acetate as the eluent to give 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl. [1]
Catalytic Applications
In the field of chemical synthesis, 2-dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl is commonly used as a ligand; it can combine with transition metals such as palladium to catalyze various conventional coupling reactions.
Sonogashira coupling
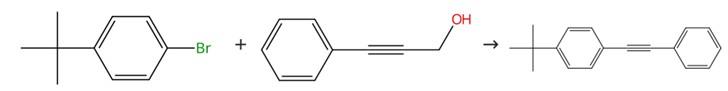
Figure2: Sonogashira coupling of 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl
A 25.0 mL Schlenk tube equipped with a magnetic stir bar was charged with the reactant (0.3 mmol, 1.5 equiv), benzene (0.2 mmol, 1.0 equiv), Pd(t-Bu₃P)₂ (0.005 mmol, 2.5 mol %), 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl (0.02 mmol, 10 mol %), KOtBu (0.5 mmol, 2.5 equiv), and THF (2.0 mL). The tube was heated at 120 °C for 14 hours, then allowed to cool to ambient temperature. Ethyl acetate (2.0 mL) was added, and the mixture was filtered. The solvents were removed under reduced pressure, and the residue was purified by column chromatography on silica gel using 100% petroleum ether as the eluent. [2]
Buchwald-Hartwig amination
An oven-dried microwave vial equipped with a stir bar was charged with Pd₂(dba)₃ (23 mg, 0.0064 mmol, 1.6 mol%), 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl (5 mg, 0.013 mmol, 3.2 equiv), 6-bromo-1-naphthol (89 mg, 0.4 mmol, 1 equiv), and N-(2-(thiophen-2-yl)ethyl)propan-1-amine (271 mg, 1.6 mmol, 4.0 equiv). The vial was capped with a Supelco aluminum crimp seal fitted with a PTFE/butyl septum, then evacuated and backfilled with argon (three cycles). Dry, degassed THF (0.6 mL) and LiHMDS (1.4 mL, 1.4 mmol, 3.5 equiv, 1 M in THF) were added to the mixture. The solution was stirred at 90 °C for 24 hours, then cooled to room temperature. The mixture was diluted with saturated aqueous NH₄Cl (10 mL) and EtOAc (10 mL), and the layers were separated. The aqueous layer was extracted with EtOAc (3 × 10 mL). The combined organic layers were washed with water (30 mL) and brine (30 mL), dried over Na₂SO₄, and evaporated. The crude product was purified by column chromatography on silica gel, eluting with n-pentane–EtOAc (15:1), to afford 2-benzyl-7-phenylnaphthalen-1-ol. [3]
Reference
[1] Xiao, Yuxuan; et al, Palladium-Catalyzed Coupling of Aryl Chlorides with Secondary Phosphines to Construct Unsymmetrical Tertiary Phosphines, Organic Letters 2024, 26, 10564-10569.
[2] Kapusniak, Lukasz; et al, A Mild One-Pot Reduction of Phosphine(V) Oxides Affording Phosphine(III) and Their Metal Catalysts, Organometallics (2021), 40(6), 693-701.
[3] Corpas, Javier; et al, Excited-state protonation and reduction enables the umpolung Birch reduction of naphthalenes, Chem 2025, 11, 102342.
You may like
See also
Lastest Price from 2-Dicyclohexylphosphino-2'-(N,N-dimethylamino)biphenyl manufacturers
US $39.00-422.00/g2025-02-08
- CAS:
- 213697-53-1
- Min. Order:
- 5g
- Purity:
- 0.98
- Supply Ability:
- 25kg

US $0.00-0.00/KG2024-12-17
- CAS:
- 213697-53-1
- Min. Order:
- 1KG
- Purity:
- 0.98
- Supply Ability:
- 500kg