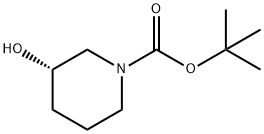
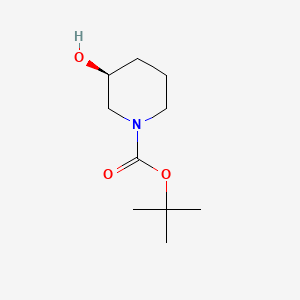
Synthetic Method and FTIR analysis of (S)-1-Boc-3-hydroxypiperidine
(S)-1-Boc-3-hydroxypiperidine is a piperidine derivative that appears as a white to tan solid powder at room temperature and under normal pressure. It can be prepared from 3-hydroxypiperidine through N-protection with Boc₂O under alkaline conditions. (S)-1-Boc-3-hydroxypiperidine serves as an important chiral intermediate in the synthesis of ibrutinib, an anticancer drug targeting B‑cell malignancies and a newly approved medication for the treatment of lymphoma.
Synthetic Method

Figure1: Synthetic method of (S)-1-Boc-3-hydroxypiperidine
In a 1.0 mL reaction system, a mixture was prepared containing 20 mM ketone (200 μL, dissolved in 10% isopropanol), cell lysate (600 μL), and 20 mM NAD(P)H (200 μL) in Na₂HPO₄–KH₂PO₄ buffer (50 mM, pH 7.0). The mixture was shaken at 200 rpm and 30 °C for 12 h, followed by extraction with ethyl acetate. The organic layer was dried over anhydrous sodium sulfate, concentrated under vacuum, and the resulting residue was purified by silica gel column chromatography to afford the target product (S)-1-Boc-3-hydroxypiperidine. The conversion rate and enantiomeric excess of the product were determined by chiral gas chromatography (GC) and high-performance liquid chromatography (HPLC) [1].
FTIR analysis
To characterize the functional groups of the biocatalyzed product, Fourier transform infrared (FTIR) analysis was performed, and the IR spectrum of (S)-1-Boc-3-hydroxypiperidine was recorded with a resolution of 4 cm⁻¹ over a range of 500–4000 cm⁻¹, accumulating 45 scans in transmission mode using a KBr pellet. The resulting spectrum exhibited characteristic peaks corresponding to (S)-1-Boc-3-hydroxypiperidine, as shown in Figure S2, with a distinct absorption band at 3462.46 cm⁻¹ clearly indicating the presence of a hydroxyl (–OH) group. Furthermore, (S)-1-Boc-3-hydroxypiperidine displayed additional prominent absorptions in the regions of 2974.18–2921.05 cm⁻¹, attributed to C–H stretching vibrations, and 1390.15–1239.36 cm⁻¹, corresponding to C–N and C–O stretching modes, thereby confirming the coexistence of hydroxyl and amide functionalities within the molecular structure. [2]
Chiral resolution and impurity quantification
The KRED mediated chiral synthesis of (S)-1-BOC-3-Hydroxypiperidine have shown to be effective approach offering green and sustainable method for the producing pharmaceutical intermediates. The application of (S) 1-BOC-3-Hydroxypiperidine in drug like ibrutinib high lights their value in pharmaceutical industries. For the chiral resolution and impurity quantification, the devel oped HPLC method gives precise, specific, sensitive and accurate analysis for the simultaneous quantitation of (R) 1-BOC-3-Hydroxypiperidine isomer in (S)-1-BOC-3-Hydroxypiperidine samples. This method had low LOD and LOQ values for (R)-1-BOC-3-Hydroxypiperidine isomer. This HPLC method has been shown to be great for quality assessment of the (S)-1-BOC-3-Hydroxypiperidine sam ple for (R)-1-BOC-3-Hydroxypiperidine isomer. There fore, it can be considered in the quality control analysis and also can be used in pharmaceutical drug substances quantification. [2]
Nucleophilic Aromatic Substitution Reaction
(S)-1-Boc-3-hydroxypiperidine (2.07 mmol) and an amine derivative (1.80 mmol) were placed in a 100 mL round-bottom flask and dissolved in 20 mL of DMSO. While stirring, potassium tert-butoxide (0.25 g, 2.25 mmol) was slowly added, causing a slight exotherm. The mixture was stirred at room temperature for 3–5 h, and reaction completion was monitored by TLC. After adding 50 mL of water and stirring for 20 min, the mixture was filtered under reduced pressure. The filter cake was washed three times with water and dried in an oven at 50 °C. Alternatively, the aqueous phase can be extracted with ethyl acetate, and (S)-1-Boc-3-hydroxypiperidine derivatives recovered from the combined organic layers after drying over anhydrous Na₂SO₄ and concentration. [3]
Pharmaceutical Applications
(S)-1-Boc-3-hydroxypiperidine is a key chiral building block widely employed in the synthesis of diverse pharmaceuticals, owing to the prevalence of piperidine rings in numerous bioactive molecules derived from both natural and synthetic origins. Piperidine, a heterocyclic amine consisting of a six-membered ring with five methylene bridges and one amine bridge, acquires a chiral center upon hydroxylation at the 3-position, a modification that can significantly influence biological activity. Consequently, (S)-1-Boc-3-hydroxypiperidine serves as an essential precursor for preparing various therapeutic agents. Notably, this compound is a crucial intermediate in the synthesis of ibrutinib, the active pharmaceutical ingredient in Imbruvica, a recently approved drug for lymphoma treatment. [2]
Reference
[1] Qin, Lei; et al, Efficient synthesis of chiral heterocyclic alcohols by designed carbonyl reductases with coenzyme regeneration system, Molecular Catalysis 2025, 586, 115430.
[2] Sharma S K, Sultana S. Chiral-selective biocatalysis of (S)-1-BOC-3-hydroxypiperidine: developing analytical method for quantifying (R)-isomer impurities[J]. Chemical Papers, 2025, 79(8): 5409-5425.
[3] Wang, Shihao; et al, Design, synthesis, and evaluation of antitumor activity of quinazoline derivatives containing different terminal segments of basic amine groups, Medicinal Chemistry Research 2025, 34, 134-153.
You may like
Related articles And Qustion
Lastest Price from (S)-1-Boc-3-hydroxypiperidine manufacturers

US $50.00/kg2025-04-21
- CAS:
- 143900-44-1
- Min. Order:
- 1kg
- Purity:
- 99
- Supply Ability:
- 5000
US $0.00-0.00/KG2025-04-04
- CAS:
- 143900-44-1
- Min. Order:
- 1KG
- Purity:
- 98%
- Supply Ability:
- 1ton