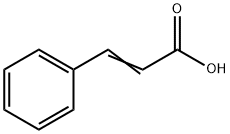
The Synthesis of Cinnamic Acid
Cinnamic acid is a natural active ingredient with diverse applications, extensively utilised in pharmaceuticals, perfumery, polymers, cosmetics, and agriculture. It can be synthesised with high yields and selectivities via Knoevenagel, Perkin, Pechman, Reformatsky, and Wittig reactions using various catalysts, alongside a range of waste reduction and recovery procedures.
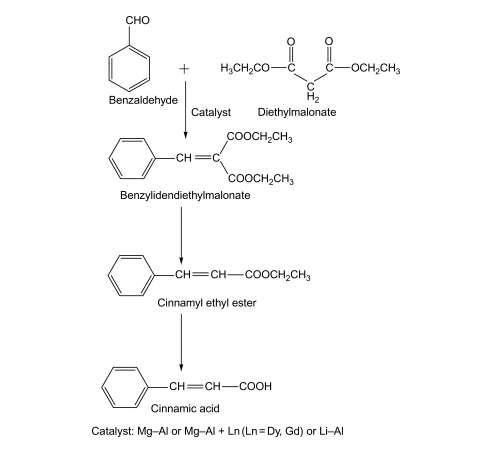
The most convenient, efficient, and environmental-friendly synthesis method for cinnamic acid is based on the Knoevenagel condensation reaction between diethylmalonate and benzaldehyde, catalysed by Mg/Al mixed oxides, Mg–Al+Ln (Ln=Dy,Gd), or Li/Al catalysts. The reaction pathway is as follows:
Other synthetic methods include the Perkin reaction. Cinnamic acid is prepared by reacting aromatic aldehydes with aliphatic carboxylic acid anhydrides in the presence of a base, particularly employing the sodium or potassium salt of the corresponding carboxylic acid anhydride as the reagent. Thus, reacting potassium acetate with benzaldehyde at 180°C for 8 hours yields cinnamic acid in 70–72% yield. Substituting sodium acetate for potassium acetate under identical conditions results in reduced yields.
You may like
Related articles And Qustion
See also
Lastest Price from Cinnamic acid manufacturers

US $0.00-0.00/kg2025-04-21
- CAS:
- 621-82-9
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 25tons/month

US $6.00/kg2025-04-21
- CAS:
- 621-82-9
- Min. Order:
- 7kg
- Purity:
- 0.99
- Supply Ability:
- 10000