Thermodynamic parameters and Animal study of Lenvatinib mesylate
Lenvatinib mesylate is an orally administered receptor-type tyrosine kinase inhibitor that was developed by Eisai and approved in 2015. It inhibits the tyrosine kinase activity associated with proangiogenic and oncogenic pathways, targeting vascular endothelial growth factor receptors (VEGFRs) 1–3, fibroblast growth factor receptors (FGFRs) 1–4, and the RET proto‑oncogene. In both in vitro and in vivo models, lenvatinib mesylate suppresses angiogenesis driven by vascular endothelial growth factor (VEGF) and fibroblast growth factor (FGF), and treatment with lenvatinib significantly inhibits tumor growth across various human cancer xenograft mouse models. This chapter provides an overview of the development of lenvatinib. In preclinical studies, Lenvatinib mesylate demonstrated inhibition of VEGF- and FGF-induced angiogenesis, and its antitumor activity against human thyroid cancer cell lines was attributed to the suppression of VEGFR, FGFR, and RET signaling. In clinical trials, lenvatinib exhibited therapeutic efficacy in differentiated thyroid cancer, renal cell carcinoma, and hepatocellular carcinoma (HCC). [1]

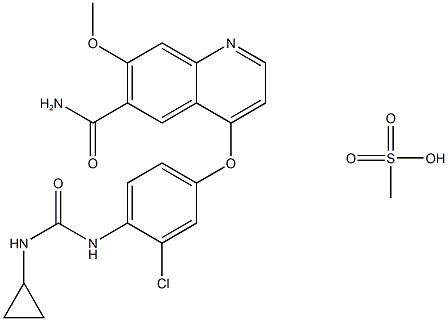
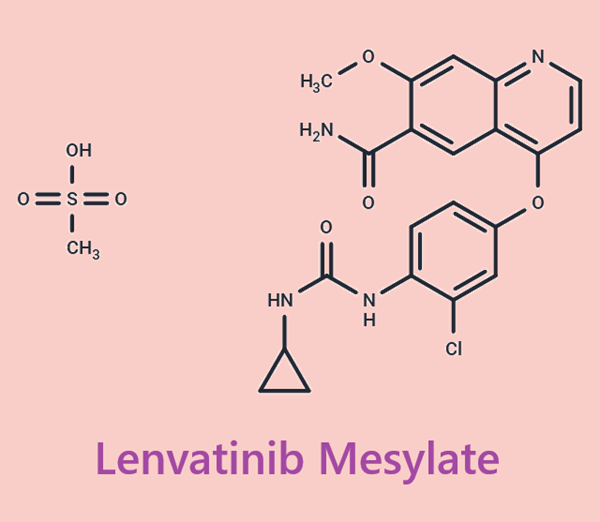
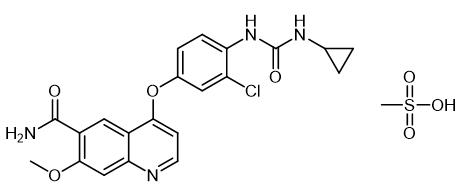
Figure1: Picture of lenvatinib mesylate
Overview
Lenvatinib mesylate is the mesylate salt form of lenvatinib, a multi-target tyrosine kinase inhibitor originally developed by Eisai Co., Ltd. of Japan. With a novel binding mode, Lenvatinib mesylate not only inhibits other pro‑angiogenic and oncogenic signaling pathway‑related tyrosine kinases involved in tumor proliferation but also selectively suppresses the kinase activity of vascular endothelial growth factor (VEGF) receptors, including VEGF‑1, VEGF‑2, and VEGF‑3. As a multi‑target tyrosine kinase inhibitor, Lenvatinib mesylate exhibits potential efficacy against various cancers. Accordingly, Eisai is conducting further studies on lenvatinib for other malignancies, such as hepatocellular carcinoma, non‑small cell lung cancer, melanoma, breast cancer, lymphoma, ovarian cancer, and others. According to data retrieved from pharmaceutical databases, there are 62 clinical trials related to lenvatinib. In July 2017, Eisai submitted marketing applications for Lenvatinib mesylate as a first‑line treatment for advanced hepatocellular carcinoma to the U.S. FDA and the European Medicines Agency. Clinical trials demonstrated that lenvatinib outperformed the current standard treatment, sorafenib, in patients with intermediate‑ to advanced‑stage hepatocellular carcinoma.
Thermodynamic parameters
At 313.15 K, the maximum solubility of lenvatinib mesylate in pure solvents was observed in methanol (4.993 × 10⁻⁴), while the lowest solubility was found in ethyl acetate (8.610 × 10⁻⁵). The mole‑fraction solubility in alcohols was higher than that in ethyl acetate, and the solubility in methanol increased approximately 9.11‑fold across the studied temperature range. In mixed solvents composed of methanol/ethanol and ethyl acetate, the solubility of lenvatinib mesylate rose with increasing mass fraction of the alcohols at a fixed temperature. Results from the KAT‑LSER model indicated that hydrogen‑bond acidity and dipolarity/polarizability exert a stronger influence on solubility. The relative average deviation (RAD) values suggested that the modified Apelblat equation and the CNIBS/R‑K model are appropriate for describing the solubility profile. Positive apparent thermodynamic parameters confirmed that the dissolution of lenvatinib mesylate is an endothermic and entropy‑driven process in all solvents examined. The apparent standard dissolution enthalpy (ΔH°sol) ranged from 39.04 kJ·mol⁻¹ to 44.26 kJ·mol⁻¹. The highest ΔG° value was observed in methanol, while the lowest was in ethyl acetate, indicating that the dissolution process is more favorable in ethyl acetate. Furthermore, the apparent standard dissolution enthalpy served as the main contributor to the Gibbs free energy during dissolution. The solubility data and model parameters obtained for lenvatinib mesylate in commonly used solvents may provide useful guidance for its purification and recrystallization. [2]
Animal study
When administered orally at a dose of 100 mg/kg, lenvatinib mesylate in combination with bevacizumab significantly inhibited local tumor growth. At the end of the treatment period, lenvatinib mesylate also markedly suppressed metastasis to regional lymph nodes and distant lungs. In an H146 xenograft model, lenvatinib mesylate inhibited H146 tumor growth in a dose-dependent manner at doses of 30 mg/kg and 100 mg/kg (BID, QD×21) and induced tumor regression at the 100 mg/kg dose. IHC analysis using an anti-CD31 antibody revealed that the 100 mg/kg dose of lenvatinib mesylate reduced microvessel density more effectively than treatment with anti-vascular endothelial growth factor antibody or imatinib. [3]
Reference
[1] Tsuruoka A, Funahashi Y, Matsui J, et al. Development of Lenvatinib Mesylate, an Angiogenesis Inhibitor Targeting VEGF and FGF Receptors[J]. Successful Drug Discovery, 2019, 4: 155-168.
[2] Chen Y, Luo Z, Ren Z, et al. The interactions and thermodynamic parameters of lenvatinib mesylate in pure and mixed solvents at several temperatures[J]. The Journal of Chemical Thermodynamics, 2023, 176: 106922.
[3] Matsui J, et al. E7080, a novel inhibitor that targets multiple kinases, has potent antitumor activities against stem cell factor producing human small cell lung cancer H146, based on angiogenesis inhibition. Int J Cancer. 2008, 122, 664-671.
You may like
Related articles And Qustion
Lastest Price from lenvatinib Mesylate manufacturers

US $0.00-0.00/KG2025-04-21
- CAS:
- 857890-39-2
- Min. Order:
- 1g
- Purity:
- 99%min
- Supply Ability:
- 10 KGS
US $0.00-0.00/kg2025-04-21
- CAS:
- 857890-39-2
- Min. Order:
- 1kg
- Purity:
- >99% by HPLC
- Supply Ability:
- 10kg/month