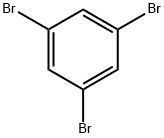
Thermodynamic Properties and Coupling reaction of 1,3,5-Tribromobenzene
1,3,5-Tribromobenzene is a polyhalogenated aromatic hydrocarbon compound, appearing as a white to brown solid powder under normal temperature and pressure. It is insoluble in water but soluble in ethyl acetate and dichloromethane. 1,3,5-Tribromobenzene exhibits similar physical and chemical properties to bromobenzene. It can be prepared via the bromination reaction of benzene with liquid bromine. This substance is primarily used as an intermediate in organic synthesis, particularly for the synthesis of multi-substituted benzene-based biologically active molecules.
Overview
Organic hydrocarbons with one or more bromine groups bonded to the carbon framework are versatile intermediates in organic synthesis. The bromo ion in hydrogen compounds is trigonally planar with 120° angles, and there are two resonance bonds so that the three H atoms are equivalent. Bromo compounds are strongly basic due to the electron-withdrawing effects of the bromine atom, both inductively and mesomerically. Historically, they have been abundant in dyes and explosives. 1,3,5-Tribromobenzene (TBB) and 1,3,5-tribromo-2,4,6-trifluorobenzene are both used as intermediates for organic compounds such as pharmaceuticals, pesticides, and dyes. Both compounds are pale yellow in color and insoluble in water. They are harmful by inhalation, in contact with skin, and if swallowed; they also irritate the eyes, respiratory system, and skin. 1,3,5-Tribromobenzene is useful as an intermediate in preparation. Haloaromatic compounds are well-known building blocks in the synthesis of pharmaceuticals and agrochemicals. Traditionally, the halogen has normally been chlorine, but bromoaromatics are assuming greater importance as the cost-effectiveness of biologically active fluorine-containing products and the synthetic value of H substituents become more widely acknowledged. [1]
Thermodynamic Properties
1,3,5-Tribromobenzene is a compound from the group of halogen-substituted benzenes. The di- and poly- substituted benzenes are candidates for the storage of thermal energy since they combine a relatively low melting point and a rather large enthalpy of fusion (heat of melting). The heat is stored in the chosen liquid mixture and is delivered upon solidification. An important part of this research consists of the experimental determination of the phase diagrams for the various binary mixtures and the semi-empirical thermodynamic description of these solid and liquid mixtures. In these descriptions, the excess functions - changes of the thermodynamic functions as a result of non-ideal mixing behaviour in the condensed phases - play an important role. If the vapour pressure is known as a function of temperature and composition of the condensed mixture, then it can be used to determine the excess functions of the condensed phases.
Photodissociation Dynamics
The photodissociation dynamics of bromobenzene,3 dibromobenzene ~o-, m-, and p-dibromobenzene!, and 1,3,5-tribromobenzene following excitation at 270 nm have been studied in molecular-beam experiments by J. Davidsson and co-workers. The results from these experiments are sum marized in Table VII. Bromobenzene, p-dibromobenzene, and tribromobenzene were observed to dissociate via only one channel, whereas two dissociation pathways were ob served in o- and m-dibromobenzene. The channel present in all the systems, assigned t1 in the table, was ascribed to predissociation from the initially excited S1 state, corre sponding to a p*←p transition in the phenyl ring, via a repulsive triplet state. The rate of this process increased with the number of bromine atoms on the ring and also with de creasing distance between the bromine atoms, as expected for a process mediated by spin±orbit interaction. In this pre vious work we did not completely understand the underlying mechanism resulting in the fast dissociation channel. [2]
Coupling reaction
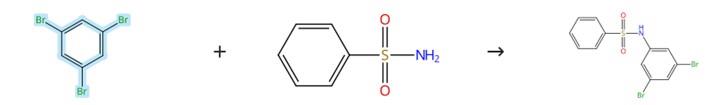
Figure1 : Coupling reaction of 1,3,5-Tribromobenzene
A mixture of benzenesulfonamide (78 mg, 0.5 mmol), 1,3,5-Tribromobenzene (236 mg, 0.75 mmol), K2CO3 (104 mg, 0.75 mmol), Fe(acac)3 (9 mg, 5 mol%), CuI (5 mg, 5 mol%), and DMSO (100 μL per 0.5 mmol scale) was heated at 130 °C under a nitrogen atmosphere in an oil bath for 15 hours. After monitoring the reaction progress by TLC, the mixture was cooled and filtered through a short column using ethyl acetate. The organic phase was then evaporated under reduced pressure, and the crude product was purified by column chromatography over silica gel (15% ethyl acetate in hexane) to afford N-(3,5-dibromophenyl)benzenesulfonamide. [3]
Reference
[1] Huinink J, Van Miltenburg J C, Oonk H A J, et al. Vapour‐pressure measurements and thermodynamic properties; 1, 3, 5‐tribromobenzene[J]. Recueil des Travaux Chimiques des Pays‐Bas, 1988, 107: 273-277.
[2] Liu Y J, Persson P, Karlsson H O, et al. Photodissociation of bromobenzene, dibromobenzene, and 1, 3, 5-tribromobenzene[J]. The Journal of chemical physics, 2004, 120: 6502-6509.
[3] Roy, Keya ; et al, Copper-Iron-Catalyzed Cross-Coupling of ortho-Substituted Sulfonamides with Sterically Hindered (Hetero)aryl Chlorides and Alkyl Halides: Synthesis of Sulfonamide Drugs, Organic Letters 2026, 28, 32-38.
You may like
Lastest Price from 1,3,5-Tribromobenzene manufacturers

US $120.00/kg2025-04-21
- CAS:
- 626-39-1
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000000

US $30.00/KG2025-04-21
- CAS:
- 626-39-1
- Min. Order:
- 1KG
- Purity:
- 98%+
- Supply Ability:
- 300MT/year