Trabectedin: use,mechanism and Drug Interactions
Introduction
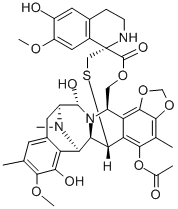
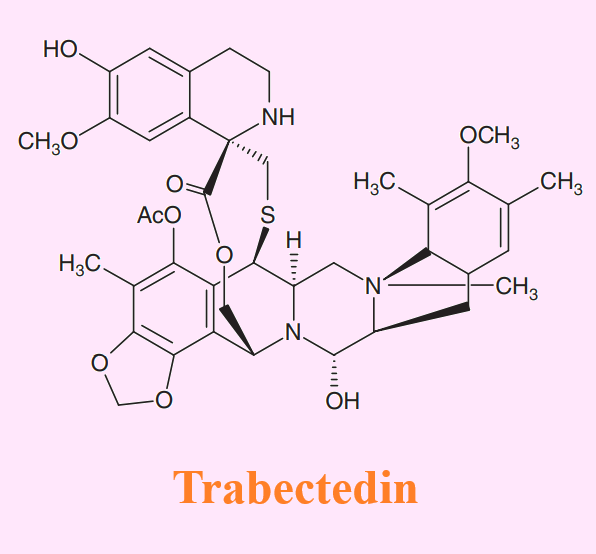
Trabectedin is a natural product that was originally isolated from the Caribbean sea squirt, Ecteinascidia turbinata but is now manufactured through an entirely synthetic route. Trabectedin is a highly potent alkylator, and its cytotoxicity is thought to be mediated through a novel interaction with DNA repair pathways. [1]
Clinical use
The recommended dose of trabectedin is 1.5 mg/m2 given intravenously over 24 hours every 21 days. Trabectedin is active in a wide range of sarcomas, particularly leiomyosarcoma and liposarcoma as well as in ovarian, breast, and prostate cancer. Trabectedin appears to have a narrow therapeutic window. Tumor responses on the phase I trials were noted at doses of 1.5mg/m2 and above, but doses above 1.5 mg/m2 were intolerable. The drug is approved in Europe for adults with advanced soft tissue sarcoma after failure of, or intolerance to, anthracyclines and ifosfamide.
Myxoid liposarcomas harboring t(12;16) are highly sensitive to trabectedin with a response rate of approximately 50%. Recent in vitro studies found that trabectedin may interfere with the binding of the characteristic FUS-CHOP fusion to promotors with subsequent cellular differentiation. Trabectedin has been evaluated in preclinical models in combination with anthracyclines, cisplatin, paclitaxel, and irinotecan. In clinical trials,combinations with gemcitabine, cisplatin, or anthracyclines have been evaluated, and a clinical trial of trabectedin in combination with docetaxel is ongoing.A phase III study in patients with relapsed ovarian cancer comparing trabectedin with liposomal doxorubicin to liposomal doxorubicin alone was recently completed and found an increase in progression-free survival in the trabectedin group (7.3 mos vs. 5.8 mos, p=0.019). This study is the basis fora new drug application to the FDA for this indication.In children, a phase I study was reported on a 3-hour infusion schedule and the recommended dose was 1.1 mg/m2.[1]
Trabectedin mechanism in tumor cells
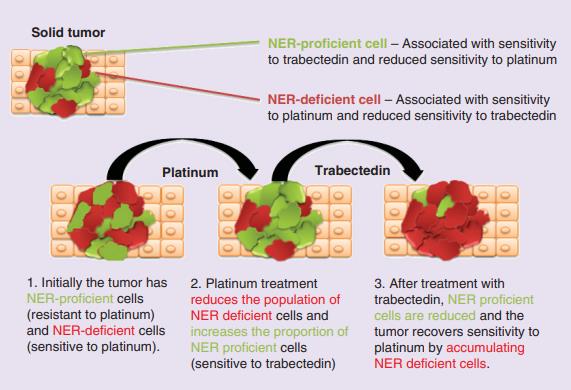
Trabectedin’s DNA-binding moiety covalently interacts with the minor groove of the DNA double helix inducing the formation of DNA adducts and bending DNA toward the major groove. The protein-binding moiety of trabectedin protrudes from the DNA doublet allowing for interaction with proteins that are recruited to the damaged site, such as XPG. This DNA excision repair protein belongs to the NER system. Formation of ternary DNA–trabectedin–XPG complexes compromises the survival of tumor cells leading to apoptosis. Platinum chemotherapy exerts its cytotoxic effect by forming DNA adducts and subsequently inhibiting DNA replication; the NER pathway is the main mechanism involved in repairing platinum DNA adducts in cellular DNA. It follows, therefore, that tumor cells with NER abnormalities (NER deficient cells) have increased sensitivity to platinum.
The direct interaction of trabectedin with components of the NER machinery inhibits the repair of specific NER substrates and, as previously described, forms a ternary complex that ultimately results in cell death, making NER-proficient cells highly sensitive to trabectedin.
Initially, a tumor has NER-proficient and NER-deficient cells. Platinum treatment is highly effective in NER deficient cells; hence, the proportion of NER-proficient cells increases after platinum therapy. The moment that the tumor is enriched with NER-proficient cells is the optimal time for trabectedin therapy as these cells are much more sensitive to trabectedin. Following exposure to trabectedin, the population of NER-deficient cells increases along with return of tumor sensitivity to platinum (Figure 1).[2]
Trabectedin mechanism in the tumor microenvironment
The tumor microenvironment operates in continuous cross-talk with tumor cells, facilitating tumor survival and progression. In the ovarian cancer microenvironment, tumor-associated macrophages increase invasiveness and promote tumor proliferation through suppressive inflammatory signals mediated by IL-6 and IL-10 and by favoring angiogenesis. IL-6 induces the proliferation, survival, migration and invasion of tumor cells. IL-6also appears to have a critical role in mediating the development of resistance to platinum through overexpression of multidrug resistance related genes and inhibition of pro-apoptotic proteins. Preclinical data demonstrated thatIL-6 blockade significantly sensitized platinum-resistant ovarian cancer cells to cisplatin. Trabectedin inhibits inflammatory mediators such as IL-6 and others. The effect of trabectedin on immune cells was demonstrated in vivo using four mouse tumor models. The seincluded three transplantable tumor models (ID8 ovarian carcinoma, MN/MCA1 fibrosarcoma and Lewis lung carcinoma) and an inducible primary fibrosarcoma. Trabectedin selectively depleted monocytes/macrophages in blood, spleens and tumors (tumor-associated macrophages) by inducing rapid apoptosis. The particular selectivity of trabectedin for monocytes over neutrophils and lymphocytes was ascribed to differential expression of signaling and decoy TNF-related apoptosis-inducing ligand receptors in these immune cells, and “provided proof-of-concept evidence for the value of macrophage targeting in anticancer therapies.”[2]
Metabolism and Elimination
Trabectedin is extensively metabolized primarily through the CYP3A4 oxidative pathway, with lesser contributions from other CYP enzymes. Fecal elimination is the primary route of excretion of trabectedin metabolites. Less than 1% of trabectedin is eliminated in stool or urine as unchanged drug. The plasma pharmacokinetics of trabectedin are highly variable but dose-proportional up to 1.8 mg/m2. Based on a population pharmacokinetic analysis, the average volume of distribution of trabectedin is 5.6 L, elimination half-life is 180 hours, and systemic clearance is 32 L/hour.
Drug Interactions
Trabectedin is primarily metabolized by CYP3A4, and inducers or inhibitors of this enzyme could alter the clearance rate of trabectedin. Ketoconazole and troleandomycin inhibit CYP3A4 metabolism of trabectedin in vitro. However,currently there are no recommendations to avoid the use of these agents or modify the dose of trabectedin when administering with other drugs that induce or inhibit CYP3A4. The systemic clearance of trabectedin is increased when coadministered with dexamethasone, but a pharmacokinetic study in adults (n =69) and a population pharmacokinetic metaanalysis of trabectedin in 603 patients concluded that relative to the variability in trabectedin clearance, this interaction was not clinically significant.[1]
References
1.Chuk MK, Balis FM, Fox E. Trabectedin. Oncologist. 2009;14(8):794-799. doi:10.1634/theoncologist.2009-0104
2.Ray-Coquard I. Trabectedin mechanism of action and platinum resistance: molecular rationale. Future Oncol. 2017;13(23s):17-21. doi:10.2217/fon-2017-0318
You may like
Related articles And Qustion
Lastest Price from Trabectedin manufacturers

US $0.00/MG2025-04-21
- CAS:
- 114899-77-3
- Min. Order:
- 1MG
- Purity:
- 98%min
- Supply Ability:
- 30kg/month

US $30.00-11.00/kg2025-03-07
- CAS:
- 114899-77-3
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 20 tons