Urapidil hydrochloride: synthesis and determination method
Introduction
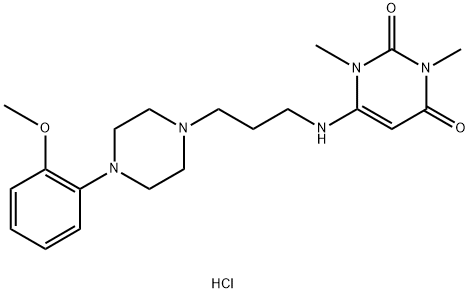
Urapidil hydrochloride is a phenyl piperazine substituted urea pyrimidine derivatives.(Figure 1). It is a highly selective α1antagonists and can induce blood vessel dilation, lower peripheral vascular resistance and reduce the feedback regulation of medulla oblongata which is called basal center of cardiovascular activity integration. Clinically, urapidil hydrochloride is a common drug that can cure heart disease, dilated cardiomyopathy, renal hypertension,acute left ventricular failure caused by kidney dialysis. At the same time, it can be safely and effectively used for the treatment of severe hypertension, hypertensive crisis and the perioperative control of hypertension.[1]

Synthesis of Urapidil hydrochloride
With 3-[(tert-butoxycarbonyl)amino]propanoic acid(2) and 1-(2-methoxyphenyl)piperazinehydrochloride(3) as starting materials, tert-butyl [3-[4-(2-methoxyphenyl)piperazin-1-yl]-3-oxopropyl]carbamate (4) was obtained by active ester method. 3-[4-(2-Methoxyphenyl)piperazin-1-yl]propylamine trihydrochloride(6hydrochloride) was prepared by the reduction of 4 followed by removing the Boc protective group. Finally, 6 was condensed with 6-chloro-1,3-dimethylpyrimidine-2,4(1H,3H)-dione (7) to obtain urapidil hydrochloride with an overall yield of above 61.2% (based on 2) and a purity of 99.95%. Urapidil hydrochloride was confirmed by HPLC, 1H NMR, 13 C NMR,MS and elemental analysis. The improved process is suitable for industrial production as it solves the technical problem of localization of key intermediate 6.[2]
The determination of urapidil hydrochloride
A number of methods have been developed for the determination of urapidi lhydrochloride, including voltammetric method, high performance liquid chromatography coupled with UV detection (HPLC), atomic absorption spectrometry (AAS), Flow-injection chemiluminescence (FI-CL) and liquid chromatography-mass spectrometry (LC-MS-MS). However, the previous methods have some drawbacks, such as high detection limits, narrow linear ranges,bad selectivity, expensive instrumentations, time-consuming procedures, which limit their practical applications. The electrochemiluminescence (ECL) has the characteristics of electrochemical analysis and chemiluminescence analysis. ECL detection coupled with capillary electrophoresis (CE) possesses the combined advantages of high sensitivity, wide linearity range, less sample volume, short analysis time, good selectivity, simple operation and low equipment cost and has been widely applied to pharmaceuticals analysis.
In Sun’s study, a simple, sensitive and selective method for determination of urapidil hydrochloride was developed using capillary electrophoresis with electrochemiluminescence (CE-ECL) technique for the first time. Under the optimized experimental conditions, the ECL intensity was linear with the concentration of urapidil hydrochloride in the range from 0.050 to 50.0 ng/mL and the detection limit was 0.014 ng/mL (S/N=3). The proposed method was used for studying pharmacokinetics of urapidil hydrochloride in rat plasma and the main pharmacokinetic parameters of the peak concentration (Cmax), half life time (T1/2) and peak concentration time (Tmax) were 240.45±21.15 ng/mL, 0.58±0.16 h and1.08±0.13 h, respectively. The recoveries of urapidil hydrochloride in the diluted extracts of rat plasma samples ranged from 96.68 to 98.82%. The RSD was lower than 3%.[2]
Determination of genotoxic impurities in urapidil hydrochloride
To establish an HPLC-MS/MS method for the determination of genotoxic impurities bis (2-chloroethyl) amine hydrochloride and O-anisidine hydrochloride in urapidil hydrochloride. Chromatographic separation was performed on a Horizon Amide 18 column (100 mm× 2.1 mm, 3 μm) using amobile phase consisting of 0.1% formic acid aqueous solution (A) and acetonitrile (B) with gradient elution. The flow rate was 0.3 mL·min−1 and the column temperature was 30 °C. Detection was carried out on a Vanquish-TS Quantis Plus triple quadrupole mass spectrometer equipped with an electrospray ionization source (ESI),operating in positive ion mode, using multiple reaction monitoring. The linear range of bis (2-chloroethyl)amine hydrochloride was 0.47-120 ng·mL-1 (r=0.999 7). The limit of detection was 0.24 ng·mL-1 , the limit of quantitation was 0.47 ng· mL-1. The recovery rates at three concentration levels ranged from 75.7% to 110.2% with RSDs of 2.9%-7.8% (n=3). The calibration curve for O-anisidine hydrochloride was linear over the range of 2.34-120 ng·mL-1 (r=0.999 9). The limit of detection was 0.59 ng·mL-1, and the limit of quantitation was 2.34 ng·mL-1.The recovery rates at three concentration levels ranged from 79.6% to 110.9% with RSDs of 1.8% to 7.9% (n=3). In three batches of urapidil hydrochloride samples, the contents of bis (2-chloroethyl) amine hydrochloride were 0.19,0.17, and 0.27 μg·g-1 , while the contents of O-anisidine hydrochloride were 1.54, 1.57, and 2.35 μg·g-1 all of which complied with the specification limit (<5.48 μg· g-1). The method is rapid, specific, sensitive and accurate, and can be effectively applied to the determination of bis (2-chloroethyl) amine hydrochloride and O-anisidine hydrochloride in urapidil hydrochloride.[3]
References
[1]Sun S, Wei Y, Long C, Deng B. Capillary electrophoresis with end-column electrochemiluminescence for ultrasensitive determination of urapidil hydrochloride in rat plasma and its application to pharmacokinetics study. J Chromatogr B Analyt Technol Biomed Life Sci. 2015;1006:146-150. doi:10.1016/j.jchromb.2015.10.011
[2]Wang D,et al.Study on the Synthetic Process of Urapidil Hydrochloride[J].Chinese Journal of Pharmaceuticals,2024,55(04):501-505.DOI:10.16522/j.cnki.cjph.2024.04.008.
[3]Zhong Ao,et al. Determination of genotoxic impurities in urapidil hydrochloride byHPLC-MS/MS method[J].Chin J Pharm Anal,2025,45(10):1826-1832.DOI:10.16155/j.0254-1793.2024-1146.
Related articles And Qustion
Lastest Price from Urapidil hydrochloride manufacturers

US $10.00/KG2025-04-21
- CAS:
- 64887-14-5
- Min. Order:
- 100KG
- Purity:
- 99%
- Supply Ability:
- 100 mt

US $0.00-0.00/kg2025-04-16
- CAS:
- 64887-14-5
- Min. Order:
- 5kg
- Purity:
- 98%-102%
- Supply Ability:
- 200kg