Usage and Biological metabolism of Ceftiofur
Ceftiofur is a third-generation cephalosporin antibiotic approved for veterinary use. First described in 1987, it is marketed by the pharmaceutical company Zoetis under the brand names Excenel, Naxcel, and Excede. It also serves as the active ingredient in Zoetis's Spectramast LC formulation for lactating cows and Spectramast DC formulation for dry cows. Ceftiofur is resistant to the antibiotic resistance enzyme beta-lactamase, and has activity against both Gram-positive and Gram-negative bacteria. Escherichia coli strains resistant to ceftiofur have been reported.

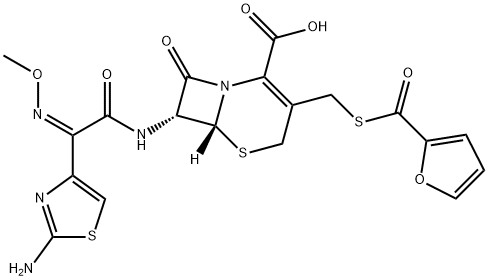
Figure1: Picture of Ceftiofur
Basic Introduction
Ceftiofur is a cell wall synthesis inhibitor that targets bacterial penicillin-binding proteins (PBPs) and exhibits anti-inflammatory effects in endotoxemia. It exerts bactericidal activity by inhibiting the synthesis of bacterial cell wall peptidoglycan, leading to bacterial cell lysis. Ceftiofur also inhibits the activation of NF-κB and MAPKs, thereby reducing the secretion of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6. A number of new cephalosporins have been developed for veterinary use, namely, ceftiofur and cefquinome. Ceftiofur does not fit clearly into the third-generation category and so has been called a ‘new generation’ cephalosporin. It has a broader Gram-positive spectrum, with good activity against Streptococcus spp., beta-lactamase-producing strains and anaerobes. Ceftiofur is rapidly metabolised to an active metabolite, desfuroylceftiofur. It has been approved for use in small animals and is indicated for urinary tract infections in dogs (2.2 mg/kg SC once daily), being active against most urinary pathogens, except P. aeruginosa (Papich, 1998). Cefquinome is not approved for use in small animals. [1]
Usage
Ceftiofur is a third-generation cephalosporin approved for use in horses by the intramuscular (IM) route only (the label dosage for streptococcal infections is 2.2 to 4.4 mg/kg intramuscularly every 24 hours). Recently, the pharmacokinetics of ceftiofur was studied after intravenous (IV) and subcutaneous (SC) administration. The IV and SC routes yielded concentrations of ceftiofur and metabolites similar to those achieved after IM administration. The SC route is convenient for use in neonatal foals that do not have IV catheters in place and is often better tolerated than the IM route. The author administers ceftiofur at dosages up to 5 to 10 mg/kg intramuscularly every 12 hours in neonatal foals, with the goal of achieving concentrations effective against bacteria that typically have higher minimum inhibitory concentration (MIC) values than Streptococcus spp and are commonly associated with neonatal sepsis, especially gram-negative enteric microbes. These dosages should not be used in adult horses because of the risk for inducing antimicrobial-associated colitis. [2]
Biological metabolism
Ceftiofur is a broad-spectrum cephalosporin that was initially developed for the treatment of bovine respiratory disease. Ceftiofur has since been approved for use in horses, swine, and poultry. Metabolism studies have shown that ceftiofur is not present in the plasma of calves one hour after administration at a dose of 1.0 mg/kg, whether given intramuscularly or intravenously. Desfuroylceftiofur is the most prevalent metabolite of ceftiofur found in plasma. In cattle, this metabolite is formed when the thioester bond of ceftiofur is cleaved, likely by circulating enzymes, to yield desfuroylceftiofur. Similar metabolic profiles have also been observed in other animal species.
In vitro activity
Ceftiofur and its primary metabolite, desfuroylceftiofur, were evaluated for in vitro activity against 539 isolates from veterinary sources. The organisms tested included Actinobacillus pleuropneumoniae, Pasteurella spp., Haemophilus somnus, Salmonella spp., Escherichia coli, staphylococci, and streptococci. Overall, ceftiofur and desfuroylceftiofur exhibited equivalent activity against the Gram-negative organisms, with all minimum inhibitory concentrations (MICs) falling within one serial dilution. Against staphylococci, MIC differences of two to three serial dilutions were observed, with an MIC₉₀ of 1.0 μg/mL for ceftiofur and 4.0–8.0 μg/mL for desfuroylceftiofur. Ceftiofur demonstrated greater activity than its metabolite against staphylococci in these comparisons. Although the MIC₉₀ obtained for Streptococcus suis for each compound was within one dilution, the MIC values against individual strains were two to three dilutions higher for desfuroylceftiofur than for ceftiofur. For bovine and equine streptococci, the MICs obtained for desfuroylceftiofur (MIC₉₀ = 0.03 μg/mL) were five serial dilutions higher than those obtained for ceftiofur (MIC₉₀ ≤ 0.001 μg/mL). [1-2]
Reference
[1] Salmon S A, Watts J L, Yancey Jr R J. In vitro activity of ceftiofur and its primary metabolite, desfuroylceftiofur, against organisms of veterinary importance[J]. Journal of Veterinary Diagnostic Investigation, 1996, 8(3): 332-336.
[2] Reppert E J. Evidence for the use of ceftiofur for treatment of metritis in dairy cattle[J]. Veterinary Clinics of North America: Food Animal Practice, 2015, 31(1): 139-149.
You may like
Related articles And Qustion
See also
Lastest Price from Ceftiofur manufacturers

US $0.00/kg2025-04-21
- CAS:
- 80370-57-6
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000kg
US $0.00-0.00/g2025-04-15
- CAS:
- 80370-57-6
- Min. Order:
- 1g
- Purity:
- 97.0%
- Supply Ability:
- 2kg