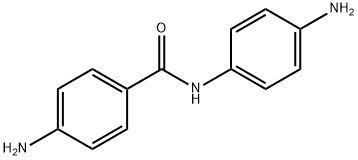
Various Applications of 4,4'-Diaminobenzanilide
4,4'-Diaminobenzanilide (DABA) is an organic compound that is used as an intermediate in the synthesis of various dyes, pigments, and other organic compounds used in the textile and printing industries. It is also used as a intermediate in the synthesis of certain types of pharmaceuticals and agrochemicals.

Inclusion complexation of 4,4′-diaminobenzanilide
Cyclodextrins (CDs) are cyclic molecules consisting of six (α-), seven (β-) or eight (γ-) glucose units correlated by α-1-4 glycosidic linkages. They have polar external surfaces and non-polar internal cavities, and can form inclusion complexes by binding small guest molecules. CDs can form different aggregates under different conditions. Many new supramolecular assemblies have been planned, such as supramolecular gels, supramolecular polymers, rotaxanes, nanospheres, and vesicles based on CD inclusion complexes. The vesicles prepared by CD inclusion complexes have gained much attention due to their importance in enthusiastic natural biomembranes, developing new biotechniques, smart materials, etc. The aim of this work was to establish a prospect of obtaining nano-inclusion complex of 4,4'-Diaminobenzanilide with α-CD and β-CD by co-precipitation method and characterised by scanning electron microscope (SEM), transmission electron microscope (TEM), FTIR, differential scanning colorimeter (DSC), powder X-ray diffraction (PXRD) and 1H NMR. The formation of DABA:CD inclusion complex has been performed by different methods. Furthermore, the spectral properties of 4,4'-Diaminobenzanilide and inclusion complexes were determined by ultraviolet–visible (UV–Vis), fluorescence and fluorescence lifetime measurements. Moreover, to explore the host–guest interaction and to study the conformational behaviour of the isolated 4,4'-Diaminobenzanilide and its complex, the inclusion complex was investigated by semi-empirical calculation in vacuum.[1]
As can be seen from the above values after inclusion complexation with DABA, a relatively weak effect was observed on the 4,4'-Diaminobenzanilide protons. H-3 and H-5 protons are located in the interior of the CD cavities and it interacts with the DABA protons. In contrast, as compared with free 4,4'-Diaminobenzanilide, the δ value of benzamido proton (H-a) shifts downfield up to 0.06 ppm in α-CD and 0.05 ppm upfield in β-CD complexes. The δ values of ‘A’ ring protons (H-b and H-c) shifted upfield up to 0.01 ppm and the ‘B’ ring protons (H-d and H-e) shifted downfield up to 0.03 and 0.05 ppm, respectively. The TEM exploration of the DABA:α-CD inclusion complex nanomaterials visualised nanochain-like agglomerates with thickness of 25–100 nm and DABA:β-CD inclusion complex nanomaterials showed spherical nanovesicular structure with a diameter of 60–120 nm. The most dominant driving force for the construction of this supramolecular structure is intermolecular hydrogen bonding and van der Waals forces. The model proposed head–head and head–tail structures of DABA:α-CD and the secondary self-assembly made the vesicular structure of DABA:β-CD. In both CDs, 4,4'-Diaminobenzanilide forms different types of 1:1 inclusion complex nanomaterials. Theoretical studies suggest that hydrophobic interaction plays an important role in determining the stability of the inclusion complexes.
Application of 4, 4′-Diaminobenzanilide Schiff Base Metal Complexes
Schiff bases are condensation products of carbonyl compound, especially aldehydes or ketones, with primary amines and they were first reported by Hugo Schiff in 1864. Several investigations on Schiff base showed that the presence of lone pair of electrons in nitrogen atom of imine group is of considerable chemical and biological applications. We can understand the coordination mode and binding nature using this docking study. It will aid in the development of a suitable treatment for the disease. In this present work we plan to synthesize novel Schiff base ligand (L) and its cobalt (II), nickel (II) and copper (II) metal complexes, which were treated with human DNA topoisomerase I and Dengue NS3 protease-helicase bi-functional enzyme by docking. The biological applications of the synthesized complexes were carried out by Cytotoxic screening analysis and DNA binding ability by using electronic spectra and Anti-Tumor activity by MTT assay. A hot solution of 1.136 g (5 mmol) 4,4'-Diaminobenzanilide in 20 mL methanol was added slowly to a hot stirring solution of 1.5112 g (10 mmol) p nitrobenzaldehyde in 20 mL methanol. The above mixture was stirred under reflux for 5 hours. On cooling to room temperature, the Schiff bases obtained are filtered, washed with diethyl ether and dried in vacuo. A unique Schiff base ligand 4-(4-nitrobenzylideneamino)-N-(4-(4-nitroobenzy lideneamino) phenyl) benzamide L has been prepared by condensing 4,4'-Diaminobenzanilide and p-nitrobenzaldehyde. The cobalt (II), nickel (II) and copper (II) complexes of L were also prepared.[2]
References
[1] N. Rajendiran, J. S., R. Sankaranarayanan. (2015). Nanochain and vesicles formed by inclusion complexation of 4,4′-diaminobenzanilide with cyclodextrins. Journal of Experimental Nanoscience, 10 1, 880–899.
[2] M. S. M. Seethalakshmi, T. P. A. T. P. A. (2020). Application of 4,4-Diaminobenzanilide Schiff Base Metal Complexes As Anti-Tumor and Anti - Dengue Agents. Journal of Water and Environmental Nanotechnology, 61 1.
You may like
Lastest Price from 4,4'-Diaminobenzanilide manufacturers

US $0.00-0.00/Kg2025-10-27
- CAS:
- 785-30-8
- Min. Order:
- 1Kg
- Purity:
- ≥99%
- Supply Ability:
- 10MT

US $0.00/KG2025-04-21
- CAS:
- 785-30-8
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month