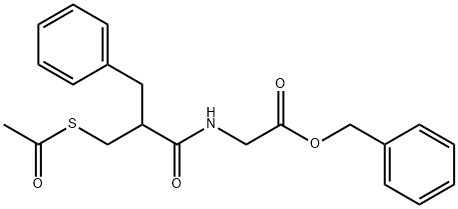
What is the mechanism of Racecadotril in treating diarrhoea?
Racecadotril, the first and only intestinal antisecretory drug, acting on the abnormal intestinal hypersecretion, decreases the loss of water and electrolytes from the gut, so reducing the dehydration risk, it increases the likelihood of home management of diarrhea. The efficacy of racecadotril has been demonstrated in all settings (inpatients, outpatients, and community-based), in patients of all ages (children, adults, and the elderly), in many countries both of high-income and low-middle-income. In addition, the safety of racecadotril has always been comparable to placebo and better than loperamide.
Mechanism
Racecadotril is a prodrug that, after oral adminis
tration, is converted into thiorphan by hydrolysis
and it exerts its effects acting on enkephalinase, a
peptidase located in the cell membrane, in differ
ent tissues, but mainly in the small intestine
mucosa. This enzyme is involved in the metabo
lism of enkephalins, endogenous opioids that in
physiologic conditions contribute to maintaining
the secretion/absorption balance in the small
intestine. In fact, enkephalines bind to δ opi
oid receptors of enterocytes, reducing the cyclic
adenosine monophosphate (cAMP) synthesis.
The final effect of this mechanism is a decrease in
both water and electrolyte secretion in the gastro
intestinal lumen.
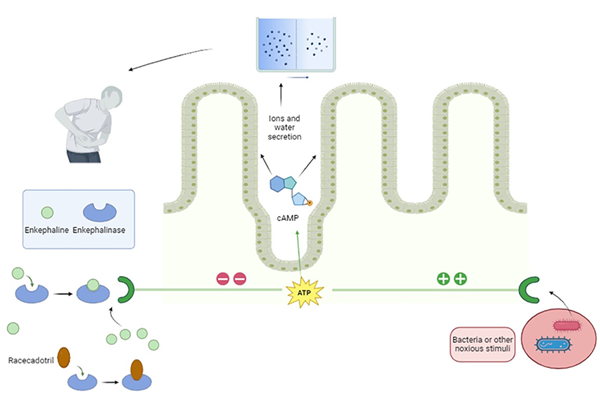
Differently from other products used in acute infectious diarrhea (AID) treatment, racecadotril, through a well-defined mechanism of action, reduces intestinal liquid loss due to the hypersecretion induced by intes tinal infections, not impacting basal secretion. Therefore, it is directly addressed to the primary pathophysiological mechanism underlying AID.
Dosage
In adults, racecadotril is administered orally,
according to the different formulations, at a total
dosage of 300–350 mg, respectively, in three or
two doses. In children, 1.5 mg/kg/dose is taken
three times daily. The maximum period of treat
ment is 1 week.
Side Effect
The most common side effect of racecadotril is headache (2/96 patients in the clinical trial by Baumer et al.). Furthermore, cutaneous/subcu taneous (including erythema, edema, urticaria, and itching have been described) and gastrointesti nal signs and symptoms have been reported. It is not futile to repeat that generally in all studies both in adults and in children adverse events incidence is not significantly different from placebo. Nevertheless, racecadotril is not indicated in anti biotic-induced diarrhea and chronic diarrhea due to a lack of clinical studies in these indications. Moreover, there are little data available on patients with severe renal or hepatic insufficiency and the drug must be administered according to the indi cations of the summary of product characteristics (SmPC). In patients with prolonged vomiting, bio availability may be reduced. Patients with heredi tary metabolic deficiencies, like galactose intolerance, Lapp-lactase deficit, or glucose-galac tose malabsorption, must avoid racecadotril.
References
[1] MARCO MANFREDI. Racecadotril in the management of diarrhea: an underestimated therapeutic option?[J]. Therapeutic Advances in Gastroenterology, 2025, 18: 17562848241310423. DOI:10.1177/17562848241310423.
You may like
Related articles And Qustion
Lastest Price from Racecadotril manufacturers

US $0.00-0.00/kg2026-03-30
- CAS:
- 81110-73-8
- Min. Order:
- 1kg
- Purity:
- 98%-102%
- Supply Ability:
- 1000KG

US $0.00-0.00/Gram2026-01-31
- CAS:
- 81110-73-8
- Min. Order:
- 1Gram
- Purity:
- USP, BP, EP, GMP
- Supply Ability:
- 100KG