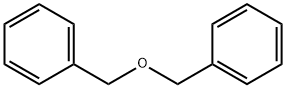
What precautions should be taken when using benzyl ether in catalytic hydrocracking?
Benzyl ether is one of the most widely used protecting groups of alcohols since it is easily installed, stable to a broad scope of reaction conditions and readily re moved in the presence of many common functionalities through hydrogenation, dissolving metal reduction, oxi dative treatments, or Lewis acid mediated cleavage. Among these, catalytic hydrocracking is the mildest method for removing the benzyl protecting group. When designing benzyl-based protecting group strategies, consideration should be given to the possibility that other oxygen-containing benzyl groups may be affected. Taking this potential drawback into account, selective deprotection reactions can be achieved through careful evaluation of the catalyst, solvent and concentration. For example, by employing a suitable two-step hydrogenolysis sequence using 10% Pd/C and Rh/Al2O3 catalysts in sequence, the hydrogenolysis of aliphatic and aromatic O-benzyl protecting groups has been successfully achieved in the presence of benzyl methyl ether.
You may like
Related articles And Qustion
See also
Lastest Price from Benzyl ether manufacturers

US $10.00/KG2025-04-21
- CAS:
- 103-50-4
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $120.00/kg2025-04-15
- CAS:
- 103-50-4
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton