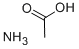ChemicalBook > Articles Catagory List >请选择分类 >why-is-an-aqueous-solution-of-ammonium-acetate-nearly-neutral
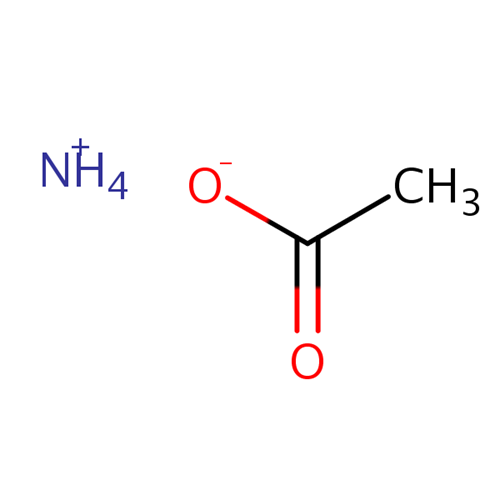
Why is an aqueous solution of ammonium acetate nearly neutral?
Apr 23,2026
Ammonium acetate is a hygroscopic, water-soluble salt of a weak acid and a weak base, commonly used as a chromatographic buffer and an auxiliary agent in organic synthesis. Its aqueous solution is nearly neutral because ammonium and acetate ions in the solution undergo simultaneous hydrolysis. Ammonium ions hydrolyze to release a small amount of hydrogen ions, while acetate ions hydrolyze to produce a small amount of hydroxide ions. The degree of hydrolysis of both is roughly equal, and the acid-base effects cancel each other out, ultimately bringing the overall solution pH close to neutral, thus exhibiting mild and stable buffering properties.
You may like
Related articles And Qustion
See also
Lastest Price from Ammonium acetate manufacturers
ammonium acetate

US $0.00-0.00/kg2026-03-11
- CAS:
- 631-61-8
- Min. Order:
- 100kg
- Purity:
- 98%
- Supply Ability:
- 20 tons per day
Ammonium acetate
US $8.80-1.10/kg2025-09-16
- CAS:
- 631-61-8
- Min. Order:
- 1kg
- Purity:
- 99%,Electronic grade(Metal impurity≤ 100ppb)
- Supply Ability:
- 500kg